Advertisements
Advertisements
Question
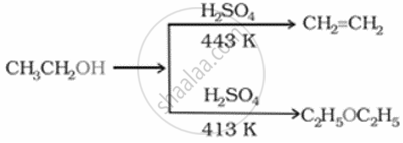
Neeta was experimenting in the lab to study the chemical reactivity of alcohol. She carried out a dehydration reaction of propanol at 140°C to 180°C. Different products were obtained at these two temperatures.
- Identify the major product formed at 140°C and the mechanism followed in this case.
- Identify the major product formed at 180°C.
Answer in Brief
Solution
- Ethanol undergoes a dehydration reaction. At 140°C, diethyl ether is formed. The formation of ether is a nucleophilic SN2 substitution bimolecular reaction.
- When the temperature exceeds 170°C, ethene is the major product. Elimination, E1 reaction.
shaalaa.com
Is there an error in this question or solution?
