Advertisements
Advertisements
Question
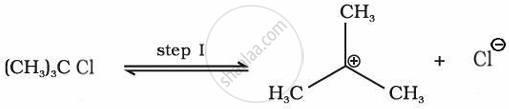
Various isomeric haloalkanes with the general formula \[\ce{C4H9Cl}\] undergo hydrolysis reactions. Among them, compound “A” is the most reactive through SN1 mechanism. Identify “A” citing the reason for your choice. Write the mechanism for the reaction.
Chemical Equations/Structures
Give Reasons
Solution
“A” is \[\ce{(CH3)3CCl}\], the carbocation intermediate obtained in tertiary alkyl halide is most stable, making A most reactive of all possible isomers.

shaalaa.com
Is there an error in this question or solution?
