Advertisements
Advertisements
Question
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Options
acetaldehyde
acetic acid
ethane
acetone
Solution
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of acetic acid
APPEARS IN
RELATED QUESTIONS
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Write IUPAC name of crotonyl alcohol.
Write the reaction to get aspirin from salicylic acid.
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
Number of oxygen atoms present in salicylaldehyde are ______.
Which alcohol is difficult to oxidise?
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Which of the following compounds is obtained, when phenol react with bromine water?
In phenols, −OH group is attached to ___________ hybridised carbon.
Phenol is obtained from cumene ____________.
Sodium metal with ethyl alcohol gives __________ gas.
Bromination of phenol, will NOT give:
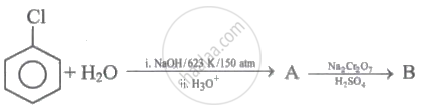
Identify 'A' and 'B' in the following series of reactions.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
+I effect of alkyl groups in alcohols increases the stability of ____________.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
Which of the following reagents is used to convert phenol to benzene?
In Raschig's method for synthesis of phenol, the reactants used are ____________.
(I)
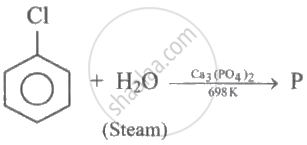
(II) \[\ce{P + Zn ->[\Delta] Q}\]
(III) \[\ce{P ->[Na2Cr2O7][H2SO4] R}\]
Q and R are respectively:
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
Name the catalyst used in commercial method of preparation of phenol.
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
Which of the following compounds has lowest boiling point?
Which among the following is not the method of preparation of phenol?
The product C in the following reaction is

What is the action of nitrous acid on aniline?
Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
