Advertisements
Advertisements
Question
Show how the following compound can be converted to benzoic acid.
Ethylbenzene
Solution
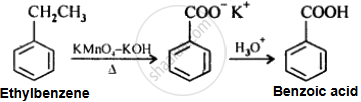
APPEARS IN
RELATED QUESTIONS
On acid hydrolysis, propane nitrile gives.
Identify ‘A' and ‘B’ in the following reaction :
C6H5MgBr + C02 `(`> ‘A’ `(PCl_5)/()`> ‘B’
Write the structures of A and B in the following reactions
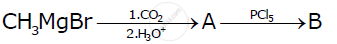
Predict the products of the following reactions:
Name the reagents used in the following reactions:

Show how the following compound can be converted to benzoic acid.
Bromobenzene
Complete the synthesis by giving missing starting material, reagent or product.
Name the reagents used in the following reactions:
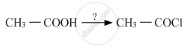
Which is the most suitable reagent for the following conversion?
\[\begin{array}{cc}
\phantom{....................}\ce{O}\phantom{.....................................}\ce{O}\phantom{.}\\
\phantom{....................}||\phantom{......................................}||\phantom{.}\\
\phantom{}\ce{CH3 - CH = CH - CH2 - C - CH3 -> CH3 - CH = CH - CH2 - C - OH}\phantom{.}
\end{array}\]
Through which of the following reactions number of carbon atoms can be increased in the chain?
(i) Grignard reaction
(ii) Cannizaro’s reaction
(iii) Aldol condensation
(iv) HVZ reaction
Match the reactions given in Column I with the suitable reagents given in Column II.
| Column I (Reactions) |
Column II (Reagents) |
| (i) Benzophenone Diphenylmethane | (a) \[\ce{LiAlH4}\] |
| (ii) Benzaldehyde 1-Phenylethanol | (b) \[\ce{DIBAL-H}\] |
| (iii) Cyclohexanone Cyclohexanol | (c) \[\ce{Zn(Hg)/Conc. HCl}\] |
| (iv) Phenyl benzoate Benzaldehyde | (d) \[\ce{CH3MgBr}\] |
Assertion: Aldehydes and ketones, both react with Tollen’s reagent to form silver mirror.
Reason: Both, aldehydes and ketones contain a carbonyl group.
Substitution of one alkyl group by replacing hydrogen of primary amines
Which of the following substance produced acetaldehyde on dry distillation?
Alkaline hydrolysis of C4H8Cl2 gives a compound (A) which on heating with NaOH and I2 produces a yellow precipitate of CHI3. The compound (A) should be ______.
A compound 'X' with molecular formula C3H8O can be oxidised to a compound 'Y' with the molecular formula C3H6O2 'X' is most likely to be ______.
Hex-4-ene-2-ol on treatment with PCC gives 'A'. 'A' on reaction with sodium hypoiodite gives 'B', which on further heating with soda lime gives 'C. The compound 'C' is ______.
