Advertisements
Advertisements
Question
State the conditions required for the given reaction to take place:
Preparation of ethyne from ethylene dibromide
Solution
The condition required for the preparation of ethylene from ethylene dibromide is that the solution should be boiled.
The chemical equation is as follows:
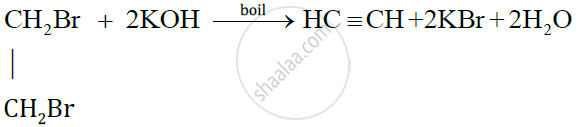
APPEARS IN
RELATED QUESTIONS
Write the structure of the following compound:
2 – methylpropane
Give the method of preparation of ethyne by 1, 2-dibromoethene.
Give a chemical test to distinguish between ethane and ethene.
Name the products formed and write an equation when ethyne is added to the iodine in an
inert solvent ?
Name the hydrocarbon which is a linear molecule.
Give reasons:
Ethyne is more reactive than ethene.
Give reason Ethene is more reactive than ethane ?
Which of the following statements is wrong about alkanes?
Give a balanced equation for the following conversion.
An alkyne to an alkene.
Substitution reactions are characteristic reactions of ______.
