Advertisements
Advertisements
Question
Which of the following statements is wrong about alkanes?
Options
They are all saturated hydrocarbons.
They can undergo addition as well as substitution reaction.
They are almost non-polar in nature.
On complete combustion, they give out carbon dioxide and water.
Solution
They can undergo addition as well as substitution reaction.
Explanation:
Alkanes only undergo substitution reactions (like halogenation). Addition reactions are shown by unsaturated hydrocarbons like alkenes and alkynes.
APPEARS IN
RELATED QUESTIONS
The compound formed where two alkyl groups are linked by 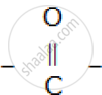 group.
group.
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulphide, ethyne]
This gas is used for welding purposes.
Write the structure of the following compound:
2 – methylpropane
Name the products formed and write an equation when ethyne is added to the chlorine in an
inert solvent ?
Name the hydrocarbon which is a linear molecule.
What is the special feature of the structure of ethyne?
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
Bromine
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
Iodine
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
hydrogen
Alkynes undergo ______ reactions.
