Advertisements
Advertisements
Question
State Raoult's law for the solution containing volatile components. What is the similarity between Raoult's law and Henry's law?
Solution
Raoult's law states that for a solution of volatile liquids, the partial vapour pressure of each component in the solution is directly proportional to its mole fraction.
Thus, if there is a solution of two liquid components (1 and 2), then for component 1:
p1 ∝ x1
\[p_1 = p_1^o x_1 \]
For component 2:
\[p_2 = p_2^o x_2 \]
Here:
p1, p2 = Partial vapour pressure of two volatile components (1 and 2) of the solution
x1 , x2 = Mole fractions of the components (1 and 2)
Similarity between Raoult's law and Henry's law:
APPEARS IN
RELATED QUESTIONS
30 g of urea (M = 60 g mol−1) is dissolved in 846 g of water. Calculate the vapour pressure of water for this solution if vapour pressure of pure water at 298 K is 23·8 mm Hg.
The vapour pressure of benzene at 30°C is 121.8 mm. By adding 15 g of non-volatile solute in 250 g of benzene, its vapour pressure is decreased to 120.2 mm. The molecular weight of solute is:
The vapour pressure of pure benzene at a certain temperature is 0.850 bar. A non-volatile, nonelectrolyte solid weighing 0.5 g is added to 39.0 g of benzene (molar mass 78 g/mol). The vapour pressure of the solution then is 0.845 bar. What is the molecular mass of the solid substance?
The vapour pressure of pure benzene at 25°C is 640 mm Hg and that of solution of solute A is 630 mm Hg. The molality of a solution is:
The solubility of a solid in a liquid is significantly affected by temperature changes.
\[\ce{Solute + Solvent -> Solution}\]
The system being in a dynamic equilibrium must follow Le-chatelier’s principle.
Considering the Le-chatelier’s principle which of the following is correct?
At a particular temperature, the vapour pressures of two liquids A and B are respectively 120 and 180 mm of mercury. If 2 moles of A and 3 moles of B are mixed to form an ideal solution, the vapour pressure of the solution at the same temperature will be (in mm of mercury):
A solution containing 10 g per dm3 of urea (molecular mass = 60 g mol–1) is isotonic with a 5% solution of a non-volatile solute. The molecular mass of this non-volatile solute is:
Two beakers of capacity 500 mL were taken. One of these beakers, labelled as “A”, was filled with 400 mL water whereas the beaker labelled “B” was filled with 400 mL of 2 M solution of NaCl. At the same temperature both the beakers were placed in closed containers of same material and same capacity as shown in figure.
At a given temperature, which of the following statement is correct about the vapour pressure of pure water and that of NaCl solution.
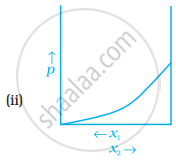
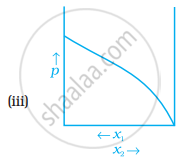
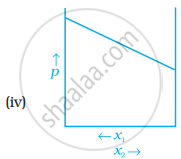
For a binary ideal liquid solution, the variation in total vapour pressure versus composition of solution is given by which of the curves?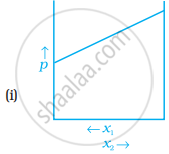
Why is the vapour pressure of an aqueous solution of glucose lower than that of water?
