Advertisements
Advertisements
Question
State the main points of Landolt’s experiment for experimental evidence of the law.
Solution
Main points of Landolt's experiment
- In an 'H' shaped tube, as shown in the figure, Landolt introduced a solution of sodium chloride \[\ce{[NaCl]}\] and silver nitrate \[\ce{[AgNO3]}\] in the limbs of the tube and sealed it and weighed it.
- The solutions were mixed by shaking to obtain a white precipitate of silver chloride.
- The tube was again weighed after the experiment.
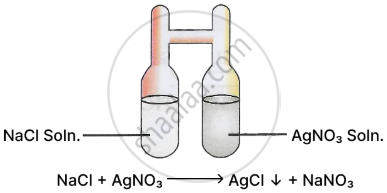
Result: Weight of H tube + reactants = Weight of H tube + products.
Hence, the total mass of reactants = total mass of products
APPEARS IN
RELATED QUESTIONS
In a reaction, 5.3 g of sodium carbonate reacted with 6 g of acetic acid. The products were 2.2 g of carbon dioxide, 0.9 g water and 8.2 g of sodium acetate. Show that these observations are in agreement with the law of conservation of mass.
Sodium carbonate + acetic acid → sodium acetate + carbon dioxide + water
Which postulate of Dalton’s atomic theory is the result of the law of conservation of mass?
State the law of conversation of mass.
What do you observe when, barium chloride solution is mixed with sodium sulphate solution ?
What is mass ?
If an object weighs 6 kg on earth. What will be its weight on moon ?
If an object is taken to the moon from the earth what will be its mass ?
State the law of conservation of mass.
Name the scientist who gave law of conservation of mass.
Name the law of chemical combination which was given by Lavoisier.
Name the law of chemical combination which was given by Proust.
State the law of conservation of mass. Give one example to illustrate this law.
Which postulate of Dalton's atomic theory can explain the law of conservation of mass ?
The law of conservation of mass was given by :
State the ‘law of conservation of mass’.
Select the correct answer from A, B, C, D & E for the statement given below:
The process of change of a liquid into vapour, [gas] on heating.
Complete the statement by filling the blank with the correct word given below.
The law of conservation of mass is strictly valid if mass and _________ are considered together.
Match the following.
| Law of conservation of mass | Sir William Crookes |
| Law of constant proportion | James Chadwick |
| Cathode rays | Joseph Proust |
| Anode rays | Lavoisier |
| Neutrons | Goldstein |
