Advertisements
Advertisements
Question
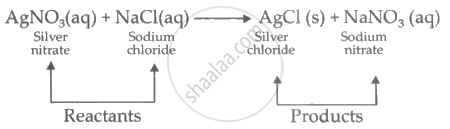
Study the following chemical reaction and answer the questions given below:
\[\ce{AgNO3_{(aq)} + NaCl_{(aq)} -> \underset{(Precipitate)}{AgCl_{(s)}}↓ + NaNO3_{(aq)}}\]
- Identify and write the type of chemical reaction.
- Write the definition of the above type of chemical reaction.
- Write the names of the reactants and products of the above reaction.
Solution
\[\ce{AgNO3_{(aq)} + NaCl_{(aq)} -> \underset{(Precipitate)}{AgCl_{(s)}}↓ + NaNO3_{(aq)}}\]
- This reaction is a double displacement reaction, in which Ag+ and Na+ displace each other from their solutions.
- The reaction in which the ions in the reactants are exchanged to form a precipitate is a double displacement reaction.
APPEARS IN
RELATED QUESTIONS
Complete the following reaction, balance it and write the name of the products CuO + HCI →_________ + _________
When the substance ‘A’ is added to a solution of BaSO4, a yellow precipitate is formed.
- What do you think substance A is likely to be?
- Name the precipitate.
- Which type of reaction is this?
Give an example of a double displacement reaction other than the one given in the following Activity.
•Take about 3 mL of sodium sulphate
solution in a test tube.
• In another test tube, take about 3 mL of
barium chloride solution.
• Mix the two solutions given in the following fig.
• What do you observe?
Write the balanced chemical equation for the following and identify the type of reaction.
\[\ce{Potassium bromide (aq) + Barium iodide (aq) -> Potassium iodide (aq) + Barium bromide (s)}\]
What is the difference between displacement and double displacement reactions? Write equations for these reactions.
What type of chemical reaction is represented by the following equation?
PQ + RS → PS + RQ
What happens when silver nitrate solution is added to sodium chloride solution?
Name the type of reaction involved.
(a) What happens when an aqueous solution of sodium sulphate reacts with an aqueous solution of barium chloride?
(b) Write the balanced chemical equation for the reaction which takes place.
(c) State the physical conditions of reactants in which the reaction will not take place.
(d) Name the type of chemical reaction which occurs.
(e) Give one example of another reaction which is of the same type as the above reaction.
Consider the reaction:
KBr (aq) + AgNO3 (aq) → KNO3 (aq) + AgBr (s)
This is an example of:
(a) decomposition reaction
(b) combination reaction
(c) double displacement reaction
(d) displacement reaction
A metal X forms a water soluble salt XNO3. When an aqueous solution of XNO3 is added to common salt solution, then a white precipitate of compound Y is formed alongwith sodium nitrate solution. Metal X is said to be the best conductor of electricity and it does not evolve hydrogen when put in dilute hydrohloric acid.
(a) What is metal X?
(b) What is salt XNO3?
(c) Name the compound Y.
(d) Write the chemical equation of the reaction which takes place on reacting XNO3 solution and common salt solution giving the physical states of all the reactants and products.
(e) What type of chemical reaction is illustrated by the above equation?
A red-brown metal X forms a salt XSO4. When hydrogen sulphide gas is passed through an aqueous solution of XSO4, then a black precipitate of XS is formed alongwith sulphuric acid solution.
(a) What could the salt XSO4 be?
(b) What is the colour of salt XSO4?
(c) Name the black precipitate XS.
(d) By using the formula of the salt obtained in (a) above, write an equation of the reaction which takes place when hydrogen sulphide gas is passed through its aqueous solution.
(e) What type of chemical reaction takes place in this case?
Fill in the blank
In the type of reaction called ....................................., ions two compounds exchange their positive and negative radicals ions respectively.
The aqueous solutions of copper sulphate and zinc sulphate appear
(A) blue and green respectively
(B) green and colourless respectively
(C) blue and brown respectively
(D) blue and colourless respectively.
(i) What is observed when a solution of potassium iodide is added to a solution of lead nitrate taken in a test tube?
(ii) What type of reaction is this?
(iii) Write a balanced chemical equation to represent the above reaction.
Give scientific reason.
It takes time for pieces of Shahabad tile to disappear in HCl, but its powder disappears rapidly.
Choose and write the correct option.
What is the type of the following reaction?
BaCl2 + ZnSO4 → BaSO4 + ZnCl2
Choose the correct option from given alternative:
When hydrogen sulphide gas is passed through a blue solution of copper sulphate, a black precipitate of copper sulphide is obtained and the sulphuric acid so formed remains in the solution. The reaction is an example of a:
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
List two types of reactions in which this reaction can be placed.
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Zinc sulphate reacts with ammonium hydroxide to give ammonium sulphate and zinc hydroxide.
Which of the following represents a precipitation reaction?
Explain the types of double displacement reactions with examples.
The following reaction is an example of:
\[\ce{4NH3_{(g)} + 5O2_{(g)} -> 4NO_{(g)} + 6H2O_{(g)}}\]
(i) displacement reaction
(ii) combination reaction
(iii) redox reaction
(iv) neutralisation reaction
Which option denotes a double displacement reaction?
Which among the following is (are) double displacement reaction(s)?
(i) \[\ce{Pb + CuCl -> PbCl2 + Cu}\]
(ii) \[\ce{Na2SO4 + BaCl2 -> BaSO4 + 2NaCl}\]
(iii) \[\ce{C + O2 -> CO2}\]
(iv) \[\ce{CH4 + 2O2 -> CO2 + 2H2O}\]
In the double displacement reaction between aqueous potassium iodide and aqueous lead nitrate, a yellow precipitate of lead iodide is formed. While performing the activity if lead nitrate is not available, which of the following can be used in place of lead nitrate?
Add potassium chromate (K2CrO4) into the solution of barium sulphate (BaSO4).
- What was the colour of the precipitate formed? Also, write the name of the precipitate.
- Write down the balanced equation for this reaction.
- Will you call this reaction a displacement reaction or a double displacement reaction? Why?

Identify the product which represents the solid state in the above reaction.
Write balanced chemical equation for the following word equation.
Lead nitrate + Potassium iodide → Lead iodide + Potassium nitrate
Is this a double displacement reaction? Justify your answer. Name the compound precipitated and write the ions present in it.
Write the method of preparation of Ca(OH)2. What happens when CO2 is passed through it? Write balanced chemical equation for the reaction involved.
Give scientific reason.
While preparing dilute sulphuric acid from concentrated sulphuric acid in the laboratory, the concentrated sulphuric acid is added slowly to water with constant stirring.
