Advertisements
Advertisements
Question
Sucrose is dextrorotatory but the mixture obtained after hydrolysis is laevorotatory. Explain.
Solution
Sucrose is dextrorotatory having [α]D = + 66.5°). On hydrolysis with dilute acids or enzymes, it gives equimolar D – (+) – glucose and D – (–) – fructose.
\[\ce{\underset{Sucrose}{C12H22O11} + H2O ->[HCl] \underset{[α]_D = + 52.4°}{\underset{D - (+) - Glucose}{C6H12O6}} + \underset{[α]_D =- 92.4°}{\underset{D - (+) - Fructose}{C6H12O6}}}\]
Since D – (–) – fructose has larger specific rotation than D – (–) – fructose has larger specific rotation than D – (+) – glucose, the resulting mixture has specific rotation of – 39.9°. Therefore, the mixture is laevorotatory.
APPEARS IN
RELATED QUESTIONS
Which one of the following is a disaccharide : Starch, Maltose, Fructose, Glucose ?
What are the products of hydrolysis of maltose?
Define the following terms:
Invert sugar
Define the following terms:
Oligosaccharides
Write the products of the following reactions:
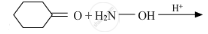
Write the products of the following reactions:
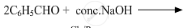
Write the products of the following reactions:
![]()
Define the following term:
Oligosaccharides
Malt sugar is obtained by the partial hydrolysis of starch by the enzyme ______.
Maltose is ______.
