Advertisements
Advertisements
Question
The compound that is not a constituent of the electrolytic mixture used in Hall-Heroult's process is ______.
Options
\[\ce{Al2O3}\]
\[\ce{NaAlO2}\]
\[\ce{Na3AlF6}\]
\[\ce{CaF2}\]
Solution
The compound that is not a constituent of the electrolytic mixture used in Hall-Heroult's process is \[\underline{\ce{NaAlO2}}\].
Explanation:
Aluminum from aluminum oxide is extracted using the Hall-Heroult technique (alumina, \[\ce{Al2O3}\]). In order to lower the melting point of alumina and increase the conductivity of the solution, an electrolytic mixture made primarily of molten alumina, a mixture of cryolite \[\ce{(Na3AlF6}\]), and additional fluxes such as calcium fluoride (\[\ce{CaF2}\]) is employed in this process.
APPEARS IN
RELATED QUESTIONS
Name the constituents of Solder.
Name the constituents of Brass.
Name the following:
The substance added along with aluminium in the Hall-Heroult's process.
How is ore purified (give equations also)
The sketch below illustrates the refin ing of aluminium by Hoope's process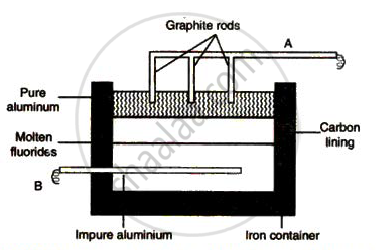
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Explain why it is preferably to use a number of graphite electrodes as anode instead of a single electrode, during the above electrolysis.
In Hoope's process, pure aluminium is collected at the ______ of the electrolytic cell.
Name the alloy used for the following purpose.
Making parts of watches
The chief ore of Aluminium is ______
Write the balanced chemical equation to show the concentration of ore in Baeyer’s process.
Sodium aluminate to aluminium hydroxide
