Advertisements
Advertisements
Question
The graph given below represents a cooling curve fore a substance being cooled from a higher temperature to a lower temperature.
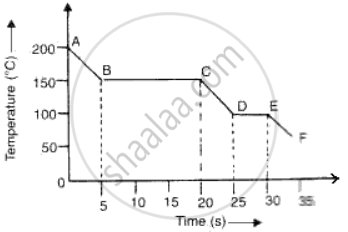
(a)What is the boiling point of the substance?
(b)What happens in the region DE?
(c)What is the melting point of the substance?
Graph
Short Note
Solution
(a)Boiling point of substance is 150°C (because the part BC represents condensation where the vapour changes into liquid without the change in temperature.
(b)DE represents freezing of the substance where the liquid changes into solid at a constant temperature of 100°C.
(c)Melting point is the temperature of the region DE where liquid changes into solid i.e., 100°C.
shaalaa.com
Is there an error in this question or solution?
