Advertisements
Advertisements
Question
The two O−O bond lengths in the ozone molecule are equal.
Solution
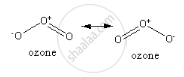
In ozone, the three oxygen atoms are arranged to form a bent shaped structure. The central oxygen atom makes a single bond with one of the terminal oxygen atoms and a double bond with the other terminal oxygen atom. But the ππ electrons of double bond are delocalised over all the three oxygen atoms. Due to which the single and the double bond are not entirely pure but are the resonance hybrids of single and double bond respectively, giving rise to the O−O bond distance as the average bond distance of the single and double bond.
The resonance structure of the ozone is given below:
APPEARS IN
RELATED QUESTIONS
Complete the equation
PbS + O3 ⟶
Account for the following :
Ozone is thermodynamically unstable.
How is O3 estimated quantitatively?
Which aerosols deplete ozone?
Give balanced equation for the following reaction:
Ozone is treated with potassium iodide solution.
Write a balanced chemical equation for the following reaction:
Ozone and lead sulphide.
Give a reason for the following :
Ozone is thermodynamically less stable than oxygen.
Explain why ozone is thermodynamically less stable than oxygen.
Which of the following is the wrong statement?
What is ozone umbrella?
