Advertisements
Advertisements
Question
Using the equation `N = N_0e^(-lambdat)` obtain the relation between half-life (T) and decay constant (`lambda`) of a radioactive substance.
Solution
A relation between half-life (T) and decay constant(`lambda`) of a radioactive substance.
`N = N_o^(e^(-lambdat))`
when t = T
`N = N_o/2`
`:. N_o/2 = N_o^(e^(-lambdaT))`
`1/2 = e^(-lambdaT)`
`2/1 = e^(lambdaT)`
`Log_e 2 = lambdaT`
`T = (log_e 2)/lambda`
`T = 0.6931/lambda`
APPEARS IN
RELATED QUESTIONS
Write symbolically the process expressing the β+ decay of `""_11^22Na`. Also write the basic nuclear process underlying this decay.
Obtain the amount of `""_27^60"Co"` necessary to provide a radioactive source of 8.0 mCi strength. The half-life of `""_27^60"Co"` is 5.3 years.
Represent Radioactive Decay curve using relation `N = N_o e^(-lambdat)` graphically
(a) Derive the relation between the decay constant and half life of a radioactive substance.
(b) A radioactive element reduces to 25% of its initial mass in 1000 years. Find its half life.
Define the activity of a given radioactive substance. Write its S.I. unit.
The radioactive isotope D decays according to the sequence
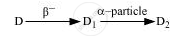
If the mass number and atomic number of D2 are 176 and 71 respectively, what is (i) the mass number (ii) atomic number of D?
57Co decays to 57Fe by β+- emission. The resulting 57Fe is in its excited state and comes to the ground state by emitting γ-rays. The half-life of β+- decay is 270 days and that of the γ-emissions is 10−8 s. A sample of 57Co gives 5.0 × 109 gamma rays per second. How much time will elapse before the emission rate of gamma rays drops to 2.5 × 109per second?
When charcoal is prepared from a living tree, it shows a disintegration rate of 15.3 disintegrations of 14C per gram per minute. A sample from an ancient piece of charcoal shows 14C activity to be 12.3 disintegrations per gram per minute. How old is this sample? Half-life of 14C is 5730 y.
A radioactive substance disintegrates into two types of daughter nuclei, one type with disintegration constant λ1 and the other type with disintegration constant λ2 . Determine the half-life of the radioactive substance.
Draw a graph showing the variation of decay rate with number of active nuclei.
