Advertisements
Advertisements
Question
Solution
Artificial radioactive substances can be produced by bombarding lighter nuclides with alpha particles, protons and neutron. The radioactive substances produces in this manner are called radioisotopes.
Radioisotopes can be used as:
(i) Rays from Radium produce satisfactorily improvement in skin diseases.
(ii) Radioactive Sulphur S35 helps to study advantages and disadvantages of fungicides.
APPEARS IN
RELATED QUESTIONS
Answer the following questions based on a hot cathode ray tube.
State the approximate voltage used to heat the filament
What are free electrons?
Give one example of isobars.
What kind of change takes place in a nucleus when a β -particle is emitted?
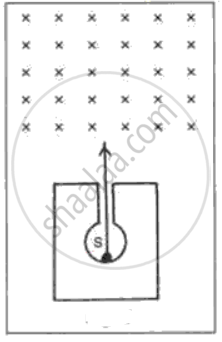
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
Draw a diagram of a simple atom showing the nucleus and electrons. In this atom:
(a) What type of charge will there be on the nucleus?
(b) What is the value of this charge?
Why should a radioactive substance not be touched by hands?
Define bound electrons.
What is meant by Radioactivity?
Which part of the atom undergoes a change in the process of radioactive decay?
