Advertisements
Advertisements
Question
What do you observe when Magnesium ribbon is burnt in oxygen.
Solution
Magnesium ribbon bums with a dazzling white light and produces a white powder which is magnesium oxide.
The reaction can be represented as
2Mg + O2 → 2MgO (white powder)
APPEARS IN
RELATED QUESTIONS
What do you observe when silver nitrate is added to a solution of sodium chloride?
How will you obtain Zinc chloride from zinc.
Also give balanced equations for the reactions
Write chemical equation for the event.
Iron filings are dropped in aqueous solution of copper sulphate.
Write the chemical equation for the event.
A reaction was brought about between ferric oxide and aluminum.
Explain the following reaction with the balanced equation.
Sulphur burns in air
Observe the following diagram and identify the type of reaction and write observation.
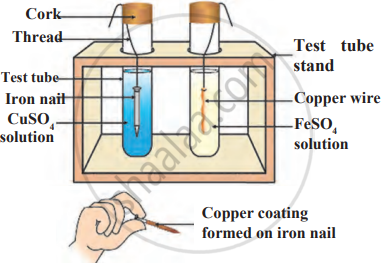
A solution of CuSO4 was kept in an iron pot. After few days the iron pot was found to have a number of holes in it. Explain the reason in terms of reactivity. Write the equation of the reaction involved.
Give the steps involved in the extraction of metals of low and medium reactivity from their respective sulphide ores.
Explain the following
- Reactivity of Al decreases if it is dipped in HNO3
- Carbon cannot reduce the oxides of Na or Mg
- NaCl is not a conductor of electricity in solid state whereas it does conduct electricity in aqueous solution as well as in molten state
- Iron articles are galvanised.
- Metals like Na, K, Ca and Mg are never found in their free state in nature.
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
