Advertisements
Advertisements
Question
What happens when a small piece of sodium is dropped in ethanol? Write the equation for this reactions.
Solution
Alcohols react with sodium leading to the evolution of hydrogen.
\[\ce{2Na + 2CH3CH2OH -> \underset{(Sodium ethoxide)}{2CH3CH2O^-Na+ + H2↑}}\]
APPEARS IN
RELATED QUESTIONS
Complete the following chemical equations :CH3COOH + Na2CO3 →
What is the common name of methanol?
Name the gas evolved when ethanoic acid is added to sodium carbonate. How would you prove the presence of this gas?
Fill in the following blank with a suitable word:
The organic acid present in vinegar is ______.
An organic compound A (molecular formula C2H4O2) reacts with Na metal to form a compound B and evolves a gas which burns with a pop sound. Compound A on treatment with an alcohol C in the presence of a little of concentrated sulphuric acid forms a sweet-smelling compound D (molecular formula C3H6O2). Compound D on treatment with NaOH solution gives back B and C. Identify A, B, C and
What do you observe when acetic acid is added to sodium bicarbonate?
A student is studying the properties of acetic acid. List two physical properties of acetic acid he observes. What happens when he adds a pinch of sodium hydrogen carbonate to this acid? Write any two observations.
Name the functional group present in the following compound:
HCOOH
Which of the following substance produces brisk effervescence with baking soda solution?
The reaction between acetic acid and sodium carbonate is shown in the following figure.
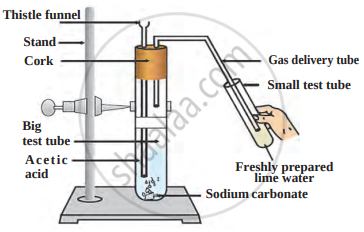
Answer the questions with the help of a diagram.
- Which gas does come out as effervescence in the big test tube?
- What is the colour change in the lime water present in the small test tube?
- Write the related reaction.
