Advertisements
Advertisements
Question
What is meant by homologous series?
Solution
A series of compounds of the same family in which each member has the same type of carbon skeleton and functional group and differs from the next member by a constant difference of one methylene group \[\ce{(-CH2-)}\] in its molecular and structural formula is called a homologous series.
APPEARS IN
RELATED QUESTIONS
Draw a formula for the first five members of the homologous series beginning with the given compound.
H–COOH
Draw a formula for the first five members of the homologous series beginning with the given compound.
CH3COCH3
Identify the functional group in the following compound.
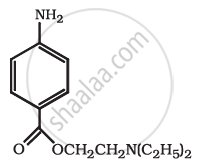
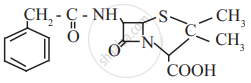
Find out all the functional groups present in the following polyfunctional compound.
Penicillin G, a naturally occurring antibiotic.
Write the first four members of the homologous series that begins with CH3CHO. Also, write down their general molecular formula.
Write the first four members of the homologous series that begins with H-C≡C-H. Also, write down their general molecular formula.
How many cyclic and acyclic isomers are possible for the molecular formula C3H6O?
Describe the classification of organic compounds based on their structure.
Write a note on homologous series.
Write the molecular formula and possible structural formula of the first four members of homologous series of carboxylic acids.
Write the molecular formula of the first six members of homologous series of nitro – alkanes.
The reagent used to confirm the presence of five hydroxyl groups in glucose is ____________.
Which of the following is a hetero-aromatic benzenoid compound?
What is the molecular formula of pyran?
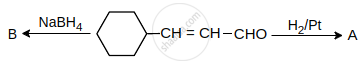
A and B are:
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{...............}\\
\ce{CH3}\phantom{..}\ce{CH3}\phantom{.............}
\end{array}\]
Identify primary, secondary, tertiary, and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3} \ce{CH3}\phantom{..............}
\end{array}\]
Identify the α - carbons in the following species and give the total number of
\[\ce{CH3 -CH2 -\overset{⊕}{C}H -CH2 -CH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{..}\ce{CH3}\phantom{..............}\\
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
Indicate the σ and π bonds in the following molecule:
\[\ce{HCONHCH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}\\
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
