Advertisements
Advertisements
Question
Solution
Let m be the mass of liquid A.
Assuming that there is no heat loss,
Heat energy given by A = Heat energy taken by B
or, m x 0.84 x ( 40 - 32) = 100 x 2.1 x ( 32 - 20)
or, m = `(100 xx 2.1 xx 12)/(0.84 xx 8)` = 375 g
APPEARS IN
RELATED QUESTIONS
Name the law on which principle of Calorimetry is based
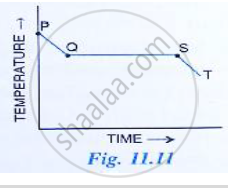
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
A block of lead of mass 250 g, at 27°C was heated in a furnace till it completely melted. Find the quantity of heat required
(a) To bring the lead block upto its melting point,
(b) To completely melt the block at its melting point (Melting point of lead is 327°C, Sp. heat cap. of lead is 0.13 J/gk and latent heat of fusion of lead is 26 J/g)
A refrigerator converts 100 g of water at 20°C to ice at -10°C in 73.5 minutes. Calculate the average rate of heat extraction from water in watt. (Sp. heat of ice= 2100 J/kgK, sp. heat of water = 4200 J/kgK, Sp. latent heat of ice = 336000 J/kg.)
40 g of ice at -16°C is dropped into water at 0°C, when 4 g of water freezes into ice. If specific heat capacity of ice is 2100 J/kg°C, what will be the latent heat of fusion of ice?
Which requires more heat: 1 g ice at 0°C or 1 g water at 0°C to raise its temperature to 10°C? Explain your answer.
Why do doctors advise to put a strip of wet cloth on the forehead of a person having high fever (temperature)?
A copper calorimeter of mass 50g contains 100g of water at 20°C. A metallic piece of mass 250 g is heated to 100°C and is then dropped into the calorimeter. The contents of the calorimeter are well stirred and its final highest temperature is recorded to be 28 °C. If the specific heat capacity of water is 4.2 J/g°C and of copper is 0.4 J/g°C, find:
(i) the heat gained by water,
(ii) the heat gained by calorimeter,
(iii) total heat supplied by the metal piece, and
(iv) the specific heat capacity of metal.
