Advertisements
Advertisements
Question
Which alkyl halide from the following pair is
- chiral
- undergoes faster SN2 reaction?
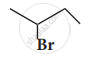
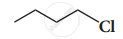
Solution
i. 
\[\begin{array}{cc}\ce{H}\phantom{..........}\\|\phantom{..........}\\\ce{CH3 - C∗ - CH2 - CH3}\\
|\phantom{..........}\\\ce{Br}\phantom{.........}\end{array}\]
Here the C* is chiral carbon atom and it is surrounded by four different groups.
ii. 
i.e., 1-chiorohutane undergoes faster SN2 reaction. CH3-CH2-CH2Cl is a primary alkyl halide and so it undergoes faster SN2 reaction.
APPEARS IN
RELATED QUESTIONS
Which of the following compounds will give racemic mixture on nucleophilic substitution by OH- ion?
- \[\begin{array}{cc}\ce{CH3 - CH - CH2Br}\\
|\phantom{....}\\\ce{C2H5}\end{array}\] - \[\begin{array}{cc}\ce{CH3}\\
|\phantom{.}\\\ce{H3C - C - C2H5}\\
|\phantom{.}\\\ce{Br}\end{array}\] - \[\begin{array}{cc}\ce{H}\\
|\phantom{.}\\\ce{CH3 - C - C2H5}\\
|\phantom{.}\\\ce{Cl}\end{array}\]
The treatment of ethyl formate with excess of RMgX gives
The carbocation formed in SN1 reaction of alkyl halide in the slow step is ______.
The raw material for Raschig process
Complete the following reaction.
\[\ce{C6H5Cl + Mg ->[THF]}\]
Complete the following reaction.
\[\ce{CHCl3 + HNO3 ->[\Delta]}\]
