Advertisements
Advertisements
Question
Which of the following compounds will give racemic mixture on nucleophilic substitution by OH- ion?
- \[\begin{array}{cc}\ce{CH3 - CH - CH2Br}\\
|\phantom{....}\\\ce{C2H5}\end{array}\] - \[\begin{array}{cc}\ce{CH3}\\
|\phantom{.}\\\ce{H3C - C - C2H5}\\
|\phantom{.}\\\ce{Br}\end{array}\] - \[\begin{array}{cc}\ce{H}\\
|\phantom{.}\\\ce{CH3 - C - C2H5}\\
|\phantom{.}\\\ce{Cl}\end{array}\]
Options
(i)
(ii) and (iii)
(iii)
(i) and (ii)
MCQ
Solution
(iii)
shaalaa.com
Classification of Organic Halogen Compounds
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
The treatment of ethyl formate with excess of RMgX gives
The carbocation formed in SN1 reaction of alkyl halide in the slow step is ______.
The raw material for Raschig process
Which alkyl halide from the following pair is
- chiral
- undergoes faster SN2 reaction?
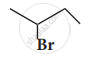
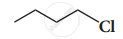
Complete the following reaction.
\[\ce{C6H5Cl + Mg ->[THF]}\]
Complete the following reaction.
\[\ce{CHCl3 + HNO3 ->[\Delta]}\]
