Advertisements
Advertisements
Question
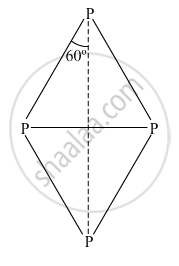
Which allotrope of phosphorus is more reactive and why?
Solution
White phosphorus is most reactive of all the allotropes of phosphorus because it is unstable due to the angular strain on P4 molecule with the bond angle of 60°.
APPEARS IN
RELATED QUESTIONS
What happens when white phosphorus is heated with concentrated NaOH solution in an inert atmosphere of CO2?
Give reasons for the following:
Red phosphorus is less reactive than white phosphorus.
Answer the following question.
Write the disproportionation reaction of H3PO3.
Which allotrope of phosphorous is most stable?
At what temperature white phosphorus changes to red phosphorus?
What is the spontaneous ignition temperature of white phosphorous?
Which of the following elements does not show allotropy?
Which of the following is correct for P4 molecule of white phosphorus?
(i) It has 6 lone pairs of electrons.
(ii) It has six P–P single bonds.
(iii) It has three P–P single bonds.
(iv) It has four lone pairs of electrons.
Phosphorus has three allotropic forms — (i) white phosphorus (ii) red phosphorus and (iii) black phosphorus. Write the difference between white and red phosphorus on the basis of their structure and reactivity.
Which one of the following is formed (mainly) when red phosphorus is heated in a sealed tube at 803 K?
