Advertisements
Advertisements
Question
Which of the following properties of atom could be explained correctly by Thomson Model of atom?
Options
Overall neutrality of atom.
Spectra of hydrogen atom.
Position of electrons, protons and neutrons in atom.
Stability of atom.
Solution
Overall neutrality of atom.
Explanation:
To the J.J Thomson model the positive charge is uniformly distributed and the electrons are embedded into it in such a manner as to give the most stable electrostatic arrangement just as watermelon of positive charge with plums or seeds (electrons) embedded into it. Thus this model is able to explain the overall neutrality of the atom.
Thomson model of atom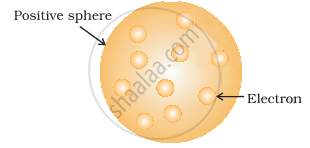
APPEARS IN
RELATED QUESTIONS
Write orbital notations for the electron in orbitals with the following quantum numbers.
n = 2, l = 1
Explain in brief, the significance of the azimuthal quantum number.
The principal quantum number (n) and magnetic quantum number (ml) for the valence electrons of rubidium atom (Z = 37) are ____________ respectively.
Total number of orbitals associated with third shell will be ______.
Calculate the total number of angular nodes and radial nodes present in 3p orbital.
The electronic configuration of valence shell of Cu is 3d104s1 and not 3d94s2. How is this configuration explained?
Match the quantum numbers with the information provided by these.
| Quantum number | Information provided |
| (i) Principal quantum number | (a) orientation of the orbital |
| (ii) Azimuthal quantum number | (b) energy and size of orbital |
| (iii) Magnetic quantum number | (c) spin of electron |
| (iv) Spin quantum number | (d) shape of the orbital |
Match the following
| (i) Photon | (a) Value is 4 for N shell |
| (ii) Electron | (b) Probability density |
| (iii) ψ2 | (c) Always positive value |
| (iv) Principal quantum number n | (d) Exhibits both momentum and wavelength |
Match species given in Column I with the electronic configuration given in Column II.
| Column I | Column II |
| (i) \[\ce{Cr}\] | (a) [Ar]3d84s0 |
| (ii) \[\ce{Fe^{2+}}\] | (b) [Ar]3d104s1 |
| (iii) \[\ce{Ni^{2+}}\] | (c) [Ar]3d64s0 |
| (iv) \[\ce{Cu}\] | (d) [Ar] 3d54s1 |
| (e) [Ar]3d64s2 |
Which of the following is the correct plot for the probability density ψ2 (r) as a function of distance 'r' of the electron from the nucleus for 2s orbitals?
