Advertisements
Advertisements
प्रश्न
Which of the following properties of atom could be explained correctly by Thomson Model of atom?
विकल्प
Overall neutrality of atom.
Spectra of hydrogen atom.
Position of electrons, protons and neutrons in atom.
Stability of atom.
उत्तर
Overall neutrality of atom.
Explanation:
To the J.J Thomson model the positive charge is uniformly distributed and the electrons are embedded into it in such a manner as to give the most stable electrostatic arrangement just as watermelon of positive charge with plums or seeds (electrons) embedded into it. Thus this model is able to explain the overall neutrality of the atom.
Thomson model of atom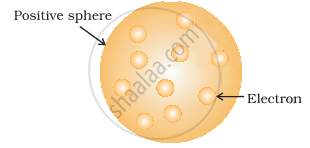
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
p-orbitals are _________ in shape.
Choose the correct option.
Principal Quantum number describes -
State Heisenberg uncertainty principle.
Give the names of quantum numbers.
Explain the anomalous behaviour of copper.
Write condensed orbital notation of electronic configuration of the following element:
Calcium (Z = 20)
Draw shapes of 2p orbitals.
Which one of the following orders is CORRECT in case of energy of the given subshells?
P: n = 4; l = 3
Q: n = 5; I = 1
R: n = 5; l = 0
S: n = 4; l = 2
The probability density plots of 1s and 2s orbitals are given in Figure:

The density of dots in a region represents the probability density of finding electrons in the region.
On the basis of above diagram which of the following statements is incorrect?
Match the following
| (i) Photon | (a) Value is 4 for N shell |
| (ii) Electron | (b) Probability density |
| (iii) ψ2 | (c) Always positive value |
| (iv) Principal quantum number n | (d) Exhibits both momentum and wavelength |
