Advertisements
Advertisements
प्रश्न
Which of the following properties of atom could be explained correctly by Thomson Model of atom?
पर्याय
Overall neutrality of atom.
Spectra of hydrogen atom.
Position of electrons, protons and neutrons in atom.
Stability of atom.
उत्तर
Overall neutrality of atom.
Explanation:
To the J.J Thomson model the positive charge is uniformly distributed and the electrons are embedded into it in such a manner as to give the most stable electrostatic arrangement just as watermelon of positive charge with plums or seeds (electrons) embedded into it. Thus this model is able to explain the overall neutrality of the atom.
Thomson model of atom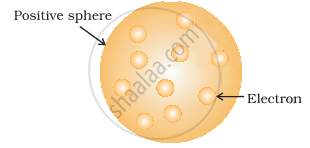
APPEARS IN
संबंधित प्रश्न
Write electronic configurations of \[\ce{Fe, Fe2+, Fe3+}\].
Write condensed orbital notation of electronic configuration of the following element:
Lithium (Z = 3)
Write condensed orbital notation of electronic configuration of the following element:
Oxygen (Z = 8)
Write condensed orbital notation of electronic configuration of the following element:
Silicon (Z = 14)
Draw shapes of 2s orbitals.
Using the concept of quantum numbers, calculate the maximum numbers of electrons present in the ‘M’ shell. Give their distribution in shells, subshells, and orbitals.
Which of the following has a greater number of electrons than neutrons?
(Mass number of Mg, C, O and Na is 24, 12, 16 and 23 respectively).
Which of the following orbitals are degenerate?
3dxy, 4dxy 3dz2, 3dyz, 4dyz, 4dz2
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, arrange the following orbitals in the increasing order of energy.
5f, 6d, 7s, 7p
Match the following species with their corresponding ground state electronic configuration.
| Atom / Ion | Electronic configuration |
| (i) \[\ce{Cu}\] | (a) 1s2 2s2 2p6 3s2 3p6 3d10 |
| (ii) \[\ce{Cu^{2+}}\] | (b) 1s2 2s2 2p6 3s2 3p6 3d10 4s2 |
| (iii) \[\ce{Zn^{2+}}\] | (c) 1s2 2s2 2p6 3s2 3p6 3d10 4s1 |
| (iv) \[\ce{Cr^{3+}}\] | (d) 1s2 2s2 2p6 3s2 3p6 3d9 |
| (e) 1s2 2s2 2p6 3s2 3p6 3d3 |
