Advertisements
Online Mock Tests
Chapters
▶ 2: Structure of Atom
3: Classification of Elements and Periodicity in Properties
4: Chemical Bonding and Molecular Structure
5: States of Matter
6: Thermodynamics
7: Equilibrium
8: Redox Reactions
9: Hydrogen
10: The s-block Elements
11: The p-block Elements
12: Organic Chemistry Some Basic Principles and Techniques
13: Hydrocarbons
14: Environmental Chemistry
![NCERT Exemplar solutions for Chemistry [English] Class 11 chapter 2 - Structure of Atom NCERT Exemplar solutions for Chemistry [English] Class 11 chapter 2 - Structure of Atom - Shaalaa.com](/images/chemistry-english-class-11_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Advertisements
Solutions for Chapter 2: Structure of Atom
Below listed, you can find solutions for Chapter 2 of CBSE NCERT Exemplar for Chemistry [English] Class 11.
NCERT Exemplar solutions for Chemistry [English] Class 11 2 Structure of Atom Multiple Choice Questions (Type - I) [Pages 14 - 23]
Which of the following conclusions could not be derived from Rutherford’s α -particle scattering experiment?
Most of the space in the atom is empty.
The radius of the atom is about 10–10 m while that of nucleus is 10–15 m
Electrons move in a circular path of fixed energy called orbits.
Electrons and the nucleus are held together by electrostatic forces of attraction.
Which of the following options does not represent ground state electronic configuration of an atom?
1s2 2s2 2p6 3s2 3p6 3d8 4s2
1s2 2s2 2p6 3s2 3p6 3d9 4s2
1s2 2s2 2p6 3s2 3p6 3d10 4s1
1s2 2s2 2p6 3s2 3p6 3d5 4s1
The probability density plots of 1s and 2s orbitals are given in Figure:

The density of dots in a region represents the probability density of finding electrons in the region.
On the basis of above diagram which of the following statements is incorrect?
1s and 2s orbitals are spherical in shape.
The probability of finding the electron is maximum near the nucleus.
The probability of finding the electron at a given distance is equal in all directions.
The probability density of electrons for 2s orbital decreases uniformly as distance from the nucleus increases.
Which of the following statement is not correct about the characteristics of cathode rays?
They start from the cathode and move towards the anode.
They travel in straight line in the absence of an external electrical or magnetic field.
Characteristics of cathode rays do not depend upon the material of electrodes in cathode ray tube.
Characteristics of cathode rays depend upon the nature of gas present in the cathode ray tube.
Which of the following statements about the electron is incorrect?
It is a negatively charged particle.
The mass of electron is equal to the mass of neutron.
It is a basic constituent of all atoms.
It is a constituent of cathode rays.
Which of the following properties of atom could be explained correctly by Thomson Model of atom?
Overall neutrality of atom.
Spectra of hydrogen atom.
Position of electrons, protons and neutrons in atom.
Stability of atom.
Two atoms are said to be isobars if.
they have same atomic number but different mass number.
they have same number of electrons but different number of neutrons.
they have same number of neutrons but different number of electrons.
sum of the number of protons and neutrons is same but the number of protons is different.
The number of radial nodes for 3p orbital is ______.
3
4
2
1
Number of angular nodes for 4d orbital is ______.
4
3
2
1
Which of the following is responsible to rule out the existence of definite paths or trajectories of electrons?
Pauli’s exclusion principle.
Heisenberg’s uncertainty principle.
Hund’s rule of maximum multiplicity.
Aufbau principle.
Total number of orbitals associated with third shell will be ______.
2
4
9
3
Orbital angular momentum depends on ______.
l
n and l
n and m
m and s
Chlorine exists in two isotopic forms, \[\ce{Cl-37}\] and \[\ce{Cl-35}\] but its atomic mass is 35.5. This indicates the ratio of \[\ce{Cl-37}\] and \[\ce{Cl-35}\] is approximately
1:2
1:1
1:3
3:1
The pair of ions having same electronic configuration is ______.
\[\ce{Cr^{3+}, Fe^{3+}}\]
\[\ce{Fe^{3+}, Mn^{2+}}\]
\[\ce{Fe^{3+}, Co^{3+}}\]
\[\ce{Sc^{3+}, Cr^{3+}}\]
For the electrons of oxygen atom, which of the following statements is correct?
Zeff for an electron in a 2s orbital is the same as Zeff for an electron in a 2p orbital.
An electron in the 2s orbital has the same energy as an electron in the 2p orbital.
Zeff for an electron in 1s orbital is the same as Zeff for an electron in a 2s orbital.
The two electrons present in the 2s orbital have spin quantum numbers ms but of opposite sign.
If travelling at same speeds, which of the following matter waves have the shortest wavelength?
Electron
Alpha particle \[\ce{(He^{2+})}\]
Neutron
Proton
Identify the pairs which are not of isotopes?
(i) 126X, 136Y
(ii) 3517X, 3717Y
(iii) 146X, 147Y
(iv) 84X, 85Y
Out of the following pairs of electrons, identify the pairs of electrons present in degenerate orbitals:
| (i) | (a) `n = 3, l = 2, m_l = -2, m_s = - 1/2` |
| (b) `n = 3, l = 2, m_l = -1, m_s = - 1/2` | |
| (ii) | (a) `n = 3, l = 1, m_l = 1, m_s = + 1/2` |
| (b) `n = 3, l = 2, m_l = 1, m_s = + 1/2` | |
| (iii) | (a) `n = 4, l = 1, m_l = 1, m_s = + 1/2` |
| (b) `n = 3, l = 2, m_l = 1, m_s = + 1/2` | |
| (iv) | (a) `n = 3, l = 2, m_l = +2, m_s = - 1/2` |
| (b) `n = 3, l = 2, m_l = +2, m_s = + 1/2` |
Which of the following sets of quantum numbers are correct?
| `n` | `l` | `m_l` | |
| (i) | 1 | 1 | +2 |
| (ii) | 2 | 1 | +1 |
| (iii) | 3 | 2 | –2 |
| (iv) | 3 | 4 | –2 |
In which of the following pairs, the ions are iso-electronic?
(i) \[\ce{Na^{+}, Mg^{2+}}\]
(ii) \[\ce{Al3^{+}, O-}\]
(iii) \[\ce{Na+ , O2-}\]
(iv) \[\ce{N3-, Cl-}\]
Which of the following statements concerning the quantum numbers are correct?
(i) Angular quantum number determines the three dimensional shape of the orbital.
(ii) The principal quantum number determines the orientation and energy of the orbital.
(iii) Magnetic quantum number determines the size of the orbital.
(iv) Spin quantum number of an electron determines the orientation of the spin of electron relative to the chosen axis.
Arrange s, p and d sub-shells of a shell in the increasing order of effective nuclear charge (Zeff) experienced by the electron present in them.
Show the distribution of electrons in oxygen atom (atomic number 8) using orbital diagram.
Nickel atom can lose two electrons to form \[\ce{Ni^{2+}}\] ion. The atomic number of nickel is 28. From which orbital will nickel lose two electrons.
Which of the following orbitals are degenerate?
3dxy, 4dxy 3dz2, 3dyz, 4dyz, 4dz2
Calculate the total number of angular nodes and radial nodes present in 3p orbital.
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, arrange the following orbitals in the increasing order of energy.
1s, 2s, 3s, 2p
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, arrange the following orbitals in the increasing order of energy.
4s, 3s, 3p, 4d
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, arrange the following orbitals in the increasing order of energy.
5p, 4d, 5d, 4f, 6s
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, arrange the following orbitals in the increasing order of energy.
5f, 6d, 7s, 7p
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, solve the questions given below:
Which of the following orbitals has the lowest energy?
4d, 4f, 5s, 5p
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, solve the questions given below:
Which of the following orbitals has the lowest energy?
5p, 5d, 5f, 6s, 6p
Which of the following will not show deflection from the path on passing through an electric field?
Proton
Cathode rays
Electron
Neutron
An atom having atomic mass number 13 has 7 neutrons. What is the atomic number of the atom?
Wavelengths of different radiations are given below:
λ(A) 300 nm, λ(B) 300 µm, λ(C) 3 nm, λ(D) = 30 A°
Arrange these radiations in the increasing order of their energies.
The electronic configuration of valence shell of Cu is 3d104s1 and not 3d94s2. How is this configuration explained?
The Balmer series in the hydrogen spectrum corresponds to the transition from n1 = 2 to n2 = 3, 4,.......... This series lies in the visible region. Calculate the wave number of line associated with the transition in Balmer series when the electron moves to n = 4 orbit. (RH = 109677 cm–1)
According to de Broglie, matter should exhibit dual behaviour, that is both particle and wave like properties. However, a cricket ball of mass 100 g does not move like a wave when it is thrown by a bowler at a speed of 100 km/h. Calculate the wavelength of the ball and explain why it does not show wave nature.
What is the experimental evidence in support of the idea that electronic energies in an atom are quantized?
Out of electron and proton which one will have, a higher velocity to produce matter waves of the same wavelength? Explain it.
A hypothetical electromagnetic wave is shown in Figure. Find out the wavelength of the radiation.
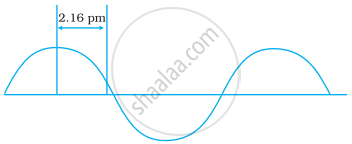
Chlorophyll present in green leaves of plants absorbs light at 4.620 × 1014 Hz. Calculate the wavelength of radiation in nanometer. Which part of the electromagnetic spectrum does it belong to?
What is the difference between the terms orbit and orbital?
Table-tennis ball has a mass 10 g and a speed of 90 m/s. If speed can be measured within an accuracy of 4% what will be the uncertainty in speed and position?
The effect of uncertainty principle is significant only for motion of microscopic particles and is negligible for the macroscopic particles. Justify the statement with the help of a suitable example.
Hydrogen atom has only one electron, so mutual repulsion between electrons is absent. However, in multielectron atoms mutual repulsion between the electrons is significant. How does this affect the energy of an electron in the orbitals of the same principal quantum number in multielectron atoms?
Match the following species with their corresponding ground state electronic configuration.
| Atom / Ion | Electronic configuration |
| (i) \[\ce{Cu}\] | (a) 1s2 2s2 2p6 3s2 3p6 3d10 |
| (ii) \[\ce{Cu^{2+}}\] | (b) 1s2 2s2 2p6 3s2 3p6 3d10 4s2 |
| (iii) \[\ce{Zn^{2+}}\] | (c) 1s2 2s2 2p6 3s2 3p6 3d10 4s1 |
| (iv) \[\ce{Cr^{3+}}\] | (d) 1s2 2s2 2p6 3s2 3p6 3d9 |
| (e) 1s2 2s2 2p6 3s2 3p6 3d3 |
Match the quantum numbers with the information provided by these.
| Quantum number | Information provided |
| (i) Principal quantum number | (a) orientation of the orbital |
| (ii) Azimuthal quantum number | (b) energy and size of orbital |
| (iii) Magnetic quantum number | (c) spin of electron |
| (iv) Spin quantum number | (d) shape of the orbital |
Match the following rules with their statements :
| Rules | Statements |
| (i) Hund's Rule | (a) No two electrons in an atom can have the same set of four quantum numbers. |
| (ii) Aufbau Principle | (b) Half-filled and completely filled orbitals have extra stability. |
| (iii) Pauli Exclusion Principle | (c) Pairing of electrons in the orbitals belonging to the same subshell does not take place until each orbital is singly occupied. |
| (d) It is impossible to determine the exact position and exact momentum of a subatomic particle simultaneously. |
|
| (iv) Heisenberg's Uncertainty Principle |
(e) In the ground state of atoms, orbitals are filled in the order of their increasing energies. |
Match the following
| (i) X-rays | (a) ν = 100 – 104 Hz |
| (ii) UV | (b) v = 1010 Hz |
| (iii) Long radio waves | (c) v = 1016 Hz |
| (iv) Microwave | (d) v = 1018 Hz |
Match the following
| (i) Photon | (a) Value is 4 for N shell |
| (ii) Electron | (b) Probability density |
| (iii) ψ2 | (c) Always positive value |
| (iv) Principal quantum number n | (d) Exhibits both momentum and wavelength |
Match species given in Column I with the electronic configuration given in Column II.
| Column I | Column II |
| (i) \[\ce{Cr}\] | (a) [Ar]3d84s0 |
| (ii) \[\ce{Fe^{2+}}\] | (b) [Ar]3d104s1 |
| (iii) \[\ce{Ni^{2+}}\] | (c) [Ar]3d64s0 |
| (iv) \[\ce{Cu}\] | (d) [Ar] 3d54s1 |
| (e) [Ar]3d64s2 |
Assertion (A): All isotopes of a given element show the same type of chemical behaviour.
Reason (R): The chemical properties of an atom are controlled by the number of electrons in the atom.
Both A and R are true and R is the correct explanation of A.
Both A and R are true but R is not the correct explanation of A.
A is true but R is false.
Both A and R are false.
Assertion (A): Black body is an ideal body that emits and absorbs radiations of all frequencies.
Reason (R): The frequency of radiation emitted by a body goes from a lower frequency to higher frequency with an increase in temperature.
Both A and R are true and R is the correct explanation of A.
Both A and R are true but R is not the explanation of A.
A is true and R is false.
Both A and R are false.
Assertion (A): It is impossible to determine the exact position and exact momentum of an electron simultaneously.
Reason (R): The path of an electron in an atom is clearly defined.
Both A and R are true and R is the correct explanation of A.
Both A and R are true and R is not the correct explanation of A.
A is true and R is false.
Both A and R are false.
What is photoelectric effect? State the result of photoelectric effect experiment that could not be explained on the basis of laws of classical physics. Explain this effect on the basis of quantum theory of electromagnetic radiations.
Threshold frequency, ν0 is the minimum frequency which a photon must possess to eject an electron from a metal. It is different for different metals. When a photon of frequency 1.0 × 1015 s–1 was allowed to hit a metal surface, an electron having 1.988 × 10–19 J of kinetic energy was emitted. Calculate the threshold frequency of this metal. Show that an electron will not be emitted if a photon with a wavelength equal to 600 nm hits the metal surface.
When an electric discharge is passed through hydrogen gas, the hydrogen molecules dissociate to produce excited hydrogen atoms. These excited atoms emit electromagnetic radiation of discrete frequencies which can be given by the general formula
`bar(v) = 109677 1/n_1^2 - 1/n_f^2`
What points of Bohr’s model of an atom can be used to arrive at this formula? Based on these points derive the above formula giving description of each step and each term.
Calculate the energy and frequency of the radiation emitted when an electron jumps from n = 3 to n = 2 in a hydrogen atom.
Why was a change in the Bohr Model of atom required? Due to which important development (s), concept of movement of an electron in an orbit was replaced by, the concept of probability of finding electron in an orbital? What is the name given to the changed model of atom?
Solutions for 2: Structure of Atom
![NCERT Exemplar solutions for Chemistry [English] Class 11 chapter 2 - Structure of Atom NCERT Exemplar solutions for Chemistry [English] Class 11 chapter 2 - Structure of Atom - Shaalaa.com](/images/chemistry-english-class-11_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
NCERT Exemplar solutions for Chemistry [English] Class 11 chapter 2 - Structure of Atom
Shaalaa.com has the CBSE Mathematics Chemistry [English] Class 11 CBSE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. NCERT Exemplar solutions for Mathematics Chemistry [English] Class 11 CBSE 2 (Structure of Atom) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. NCERT Exemplar textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry [English] Class 11 chapter 2 Structure of Atom are Discovery of Electron, Charge to Mass Ratio of Electron, Charge on the Electron, Discovery of Protons and Neutrons, Atomic Model, J. J. Thomson’s Atomic Model, Lord Rutherford’s Atomic model, Atomic Number (Z), Mass Number (A), and Number of Neutrons (n), Isobars, Isotopes, Drawbacks of Rutherford Atomic Model, Wave Nature of Electromagnetic Radiation, Particle Nature of Electromagnetic Radiation: Planck's Quantum Theory of Radiation, Evidence for the Quantized Electronic Energy Levels - Atomic Spectra, Dual Behaviour of Matter: De Broglie's relationship, Heisenberg’s Uncertainty Principle, Quantum Mechanical Model of the Atom - Orbitals and Quantum Numbers, Quantum Mechanical Model of the Atom - Concept of Shells and Subshells, Quantum Mechanical Model of the Atom - Shapes of Atomic Orbitals, Quantum Mechanical Model of the Atom - Energies of Orbitals, Quantum Mechanical Model of the Atom - Filling of Orbitals in Atom, Quantum Mechanical Model of the Atom - Electronic Configuration of Atoms, Quantum Mechanical Model of the Atom - Stability of Completely Filled and Half Filled Subshells, Atomic Mass, Bohr’s Model for Hydrogen Atom, Quantum Mechanical Model of Atom, Electromagnetic Waves : Numericals, Structure of Atom Numericals.
Using NCERT Exemplar Chemistry [English] Class 11 solutions Structure of Atom exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in NCERT Exemplar Solutions are essential questions that can be asked in the final exam. Maximum CBSE Chemistry [English] Class 11 students prefer NCERT Exemplar Textbook Solutions to score more in exams.
Get the free view of Chapter 2, Structure of Atom Chemistry [English] Class 11 additional questions for Mathematics Chemistry [English] Class 11 CBSE, and you can use Shaalaa.com to keep it handy for your exam preparation.
