Advertisements
Advertisements
Question
Which of the following solutions of KCl will have the highest value of molar conductivity?
Options
0.01 M
1 M
0.5 M
0.1 M
Solution
0.01 M
Explanation:
The relationship between specific conductivity and molar conductivity is:
Molar conductivity, ∧m = `(κ xx 1000)/"c"` cm3L–1
From the above relation, ∧m is inversely proportional to the molarity of the solution.
Therefore, the lesser the value of molarity, the more will be the molar conductivity.
RELATED QUESTIONS
State Kohlrausch’s law of independent migration of ions.
Define the following terms :
Limiting molar conductivity
Assertion: Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason: For weak electrolytes degree of dissociation increases with dilution of solution.
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
Consider figure and answer the question to given below.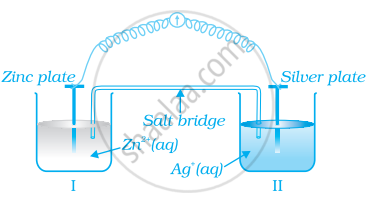
How will the concentration of Zn2+ ions and Ag+ ions be affected after the cell becomes ‘dead’?
Which of the following halogen acids is the strongest reducing agent?
The molar conductivity of 0.007 M acetic acid is 20 S cm2 mol−1. What is the dissociation constant of acetic acid? Choose the correct option.
`[(Λ_("H"^+)^ο = 350 "S" "cm"^2 "mol"^-1), (Λ_("CH"_3"COO"^-)^ο = 50 "S" "cm"^2 "mol"^-1)]`
Which of the following solutions will have the highest conductivity at 298 K?
The resistance of a conductivity cell with a 0.1 M KCl solution is 200 ohm. When the same cell is filled with a 0.02 M NaCl solution, the resistance is 1100 ohm. If the conductivity of 0.1 M KCl solution is 0.0129 ohm-1 cm-1, calculate the cell constant and molar conductivity of 0.02 M NaCl solution.
The solution of two electrolytes A and B are diluted. ^m of B increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Give a reason.
