Advertisements
Advertisements
प्रश्न
Which of the following solutions of KCl will have the highest value of molar conductivity?
पर्याय
0.01 M
1 M
0.5 M
0.1 M
उत्तर
0.01 M
Explanation:
The relationship between specific conductivity and molar conductivity is:
Molar conductivity, ∧m = `(κ xx 1000)/"c"` cm3L–1
From the above relation, ∧m is inversely proportional to the molarity of the solution.
Therefore, the lesser the value of molarity, the more will be the molar conductivity.
संबंधित प्रश्न
Resistance of conductivity cell filled with 0.1 M KCl solution is 100 ohms. If the resistance of the same cell when filled with 0.02 M KCl solution is 520 ohms, calculate the conductivity and molar conductivity of 0.02 M KCl solution. [Given: Conductivity of 0.1 M KCl solution is 1.29 S m-1 .]
Define the following terms: Molar conductivity (⋀m)
Define the following terms :
Limiting molar conductivity
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2) following curves are obtained for two electrolytes A and B : 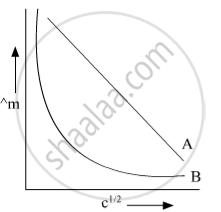
Answer the following:
(i) predict the nature of electrolytes A and B.
(ii) What happens on the extrapolation of ∧m to concentration approaching for electrolytes A and B?
\[\ce{Λ^0_m}_{(NH_4OH)}\] is equal to ______.
Assertion: `"E"_("Ag"^+ //"Ag")` increases with increase in concentration of Ag+ ions.
Reason: `"E"_("Ag"^+ //"Ag")` has a positive value.
Which of the following halogen acids is the strongest reducing agent?
Molar conductivity of substance “A” is 5.9 × 103 S/m and “B” is 1 × 10–16 S/m. Which of the two is most likely to be copper metal and why?
The unit of molar conductivity is ______.
The solution of two electrolytes A and B are diluted. ^m of B increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Give a reason.
