Advertisements
Advertisements
Question
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Solution
Dextro and laevorotatory isomers of Butan-2-ol are enantiomers of each other and both have the same boiling point and hence they cannot be separated by fractional distillation.
APPEARS IN
RELATED QUESTIONS
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
Define racemic mixture.
Which of the following halide is 2°?
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
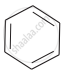
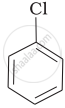
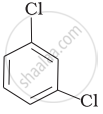
Arrange the following compounds in the increasing order of their densities.
(a)
(b)
(c)
(d)
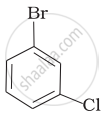
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
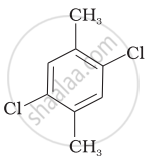 |
| (III) | 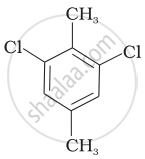 |
Assertion: The boiling points of alkyl halides decrease in the order:
\[\ce{RI > RBr > RCl > RF}\]
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
