Advertisements
Advertisements
Question
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Solution
The reaction of aniline with nitrous acid at 273 - 278 K produces benzene diazonium chloride. The reaction of sodium nitrite with hydrochloric acid produces nitrous acid in the reaction mixture. Diazotisation is the process of converting primary aromatic amines into diazonium salts. Because of its instability, the diazonium salt is generally not used immediately after preparation.
The crystalline solid benzene diazonium chloride is colorless.
It is easily soluble in water and stable at room temperature but it reacts with water when warmed.
In the dry state, it decomposes easily.
APPEARS IN
RELATED QUESTIONS
Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
Why is aniline soluble in aqueous HCl?
How will you carry out the following conversions?
toluene `->` p-toluidine
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Aniline when treated with cone. HNO3 gives
Benzene diazonium chloride is a ______.
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
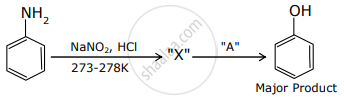
Major Product In the above chemical reaction, intermediate "X" and reagent/condition "A" are:
Aniline dissolved in dilute HCl is reacted with sodium nitrate at 0 °C. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is:
Identify A and B for the following reaction:

What are polymers?
