Advertisements
Advertisements
Question
With a labelled diagram describe in brief an activity to show the formation of ester.
Solution
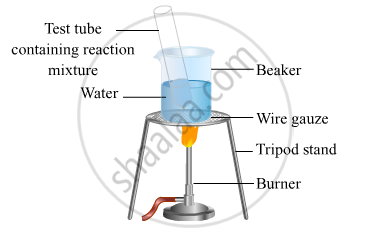
The activity given below shows the formation of ester :
- Take 1 mL ethanol (absolute alcohol) and 1 mL glacial acetic acid along with a few drops of concentrated sulphuric acid in a test tube.
- Warm in a water-bath for at least five minutes as shown in the figure below.
- Pour into a beaker containing 20-50 mL of water.
- After some time, there will be the formation of a fruity smell compound. The compound formed is an ester.
The formation of the ester can be represented by the chemical reaction as:
APPEARS IN
RELATED QUESTIONS
Ethanoic acid has a .................... odour.
(a) Rotten eggs
(b) Pungent
(c) Vinegar-like
(d) Mild
Complete the following chemical equations :CH3COOH+NaOH→
Give a reason for Conductivity of dilute hydrochloric acid is greater than that of acetic acid
An organic compound X of molecular formula C2H4O2 gives brisk effervescence with sodium hydrogen carbonate. Give the name and formula of X.
Name the product formed and give an appropriate chemical equation for the following:
Ethanol oxidised by acidified potassium dichromate?
What is glacial acetic acid ?
Which one of the following are the correct observations about acetic acid?
(A) It turns blue litmus red and smells like vinegar
(B) It turns blue litmus red and smells like burning sulphur
(C) It turns res litmus blue and smells like vinegar
(D) It turns red litmus blue and has a fruity smell
Give balanced chemical equations for the following conversion:
Calcium carbide to ethyne
A few drops of ethanoic acid were added to solid sodium carbonate. The observation made was that ______.
In the presence of the acid catalyst, ethanoic acid reacts with ethanol and ______ ester is produced.
