Advertisements
Advertisements
प्रश्न
With a labelled diagram describe in brief an activity to show the formation of ester.
उत्तर
The activity given below shows the formation of ester :
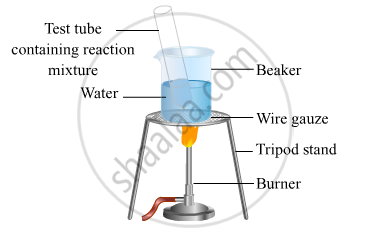
- Take 1 mL ethanol (absolute alcohol) and 1 mL glacial acetic acid along with a few drops of concentrated sulphuric acid in a test tube.
- Warm in a water-bath for at least five minutes as shown in the figure below.
- Pour into a beaker containing 20-50 mL of water.
- After some time, there will be the formation of a fruity smell compound. The compound formed is an ester.
The formation of the ester can be represented by the chemical reaction as:
\[\ce{\underset{\text{(Ethanoic acid)}}{CH3COOH} + \underset{\text{(Ethanol)}}{C2H5OH}->[conc. H2SO4] \underset{\text{(Ester)}}{CH3COOC2H5} + H2O}\]
APPEARS IN
संबंधित प्रश्न
What will you observe when dilute ethanoic acid and dilute hydrochloric acid are put on universal indicator paper, one by one? What does it show?
When ethanoic acid reacts with sodium hydrogen carbonate, then a salt X is formed and a gas Y is evolved. Name the salt X and gas. Y Describe an activity with the help of a labelled diagram of the apparatus used to prove that the evolved gas is the one which you have named. Also write the chemical equation of the reaction involved.
A neutral organic compound is warmed with some ethanoic acid and a little of conc. H2SO4. Vapours having sweet smell (fruity smell) are evolved. What type of functional group is present in this organic compound?
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is added to alcohol to denature it?
An organic acid X is a liquid, which often freezes during winter time in cold countries, having the molecular formula C2H4O2. On warming it with methanol in the presence of a few drops of concentrated sulphuric acid, a compound Y with a sweet smell is formed.
(a) Identify X and Y. Also write their formulae showing the functional group present in them.
(b) Write a chemical equation for the reaction involved.
On adding NaHCO3 to acetic acid, a gas is evolved which turns lime water milky due to the formation of:
(1) Calcium bicarbonate
(2) Calcium hydroxide
(3) Calcium carbonate
(4) Calcium acetate
Rewrite the following statement by selecting the correct option.
The formula of ethanoic acid is _____________.
Give the balanced chemical equation of the following reaction:
Evolution of carbon dioxide by the action of ethanoic acid with NaHCO3.
When ethanoic acid is treated with NaHCO^ the gas evolved is ______.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
