Advertisements
Advertisements
प्रश्न
With a labelled diagram describe in brief an activity to show the formation of ester.
उत्तर
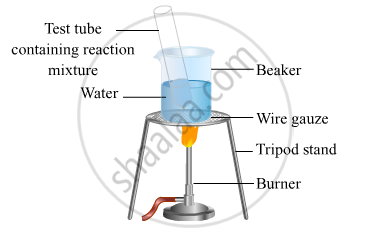
The activity given below shows the formation of ester :
- Take 1 mL ethanol (absolute alcohol) and 1 mL glacial acetic acid along with a few drops of concentrated sulphuric acid in a test tube.
- Warm in a water-bath for at least five minutes as shown in the figure below.
- Pour into a beaker containing 20-50 mL of water.
- After some time, there will be the formation of a fruity smell compound. The compound formed is an ester.
The formation of the ester can be represented by the chemical reaction as:
\[\ce{\underset{\text{(Ethanoic acid)}}{CH3COOH} + \underset{\text{(Ethanol)}}{C2H5OH}->[conc. H2SO4] \underset{\text{(Ester)}}{CH3COOC2H5} + H2O}\]
APPEARS IN
संबंधित प्रश्न
While studying saponification reaction, a student measures the temperature of the reaction mixture and also finds its nature using blue/red litmus paper. On the basis of his observations the correct conclusion would be
(A) the reaction is exothermic and the reaction mixture is acidic.
(B) the reaction is endothermic and the reaction mixture is acidic.
(C) the reaction is endothermic and the reaction mixture is basic.
(D) the reaction is exothermic and the reaction mixture is basic.
Identify the term or substance based on the descriptions given below
Ice like crystals formed on cooling an organic acid sufficiently.
When zinc powder is added to acetic acid ______________
(a) the mixture becomes warm
(b) a gas is evolved
(c) the colour of the mixture becomes yellow
(d) a solid settles at the bottom
What happens when propanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid? Write equation of the reaction involved.
What will you observe when dilute ethanoic acid and dilute hydrochloric acid are put on universal indicator paper, one by one? What does it show?
What do you observe when acetic acid is added to ethyl alcohol in the presence of sulphuric acid?
A student takes Na2CO3 powder in a test tube and pours some drops of acetic acid over it. He observes:
(A) no reaction in the test tube
(B) colourless gas with pungent smell
(C) bubbles of a colourless and odourless gas
(D) white fumes with smell of vinegar
What is thr boilng point of acetic acid?
Identify the term or substance based on the descriptions given below:
Ice like crystals formed on cooling an organic acid sufficiently.
A student while observing the properties of acetic acid would report that this smells like ______.
