Advertisements
Advertisements
Question
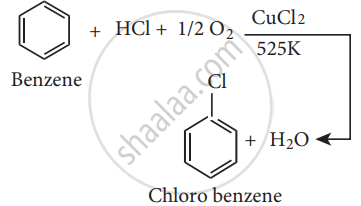
Write a short note on raschig process.
Solution
Chloro benzene is commercially prepared by passing a mixture of benzene vapour, air and HCl overheated cupric chloride, this reaction is called the Raschig process,
APPEARS IN
RELATED QUESTIONS
C -X bond is strongest in ______.
Freon-12 is manufactured from tetrachloro methane by ______.
The major products obtained when chlorobenzene is nitrated with HNO3 and con H2SO4
What happens when chloroform reacts with oxygen in the presence of sunlight?
Write down the possible isomers of C5H11Br and give their IUPAC and common names.
Reagents and the conditions used in the reactions are given below. Complete the table by writing down the product and the name of the reaction.
| Reaction | Product | Name of the reaction |
| \[\ce{CH3CH2OH + SOCl2 ->[pyridine] ?}\] | ______ | ______ |
| \[\ce{CH3CH2Br + AgF ->}\] ? | ______ | ______ |
| \[\ce{C6H5Cl + Na ->[Ether]}\] | ______ | ______ |
Discuss the aromatic nucleophilic substitutions reaction of chlorobenzene.
Write a short note on Darzen’s process.
In aryl halide, halogen atom is attached ______.
