Advertisements
Advertisements
Question
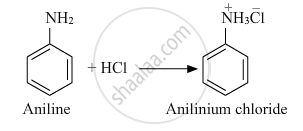
Write the chemical equations involved when aniline is treated with the following reagents: HCI
Solution
RELATED QUESTIONS
Arrange the following:
Aniline, p-nitroaniline, p-methylaniline - in the increasing order of their basic strength
Arrange the following:
In increasing order of basic strength:
Aniline, p-nitroaniline and p-toluidine
Write a short note on the following:
Carbylamine reaction
Write the structures of the main products of the following reactions:
Arrange the following in decreasing order of their basic strength:
C6H5NH2, C2H5NH2, (C2H5)2NH2, NH3
The correct decreasing order of basic strength of the following species is ______.
\[\ce{H2O, NH3, OH-, NH^{-}2}\]
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
Which of the following is most basic?
What is the correct decreasing order of the basic character of the three amines and ammonia?
The correct order of the increasing basic nature of Ammonia, Methylamine and Aniline is:
