Advertisements
Advertisements
Question
Write down the number of neutrons in the nucleus of an atom having atomic number 17 and mass number 37.
Solution
A = n + p (At. No.) A is mass number
37 = n + 17
Number of neutrons = n = 37 – 17 = 20
APPEARS IN
RELATED QUESTIONS
Compare the properties of electrons, protons and neutrons.
Name the subatomic particle whose relative charge is : 0
Fill in the blank of the following statement :
If the nucleus of an atom has atomic number 17, mass number 37 and there are 17 electrons outside the nucleus, the number of neutrons in it is __________.
Fill in the blank of the following statement :
The subatomic particle not present in a hydrogen atom is ________
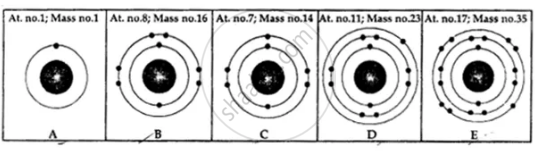
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
The number of neutrons present in 73Li is ______.
On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
Isotopes exist because atoms of the same element can have different numbers of ______.
What is the charge of a neutron?
