Advertisements
Advertisements
Question
Write notes on Nuclear fission
Solution
Nuclear fission:
1) The process of splitting a heavy nucleus into two lighter nuclei after bombardment with neutrons is called nuclear fission.
2) The process of nuclear fission was first discovered by German scientists Otto Hahn and Strassman in 1939.
3) Example of nuclear fission:
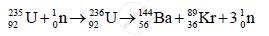
In the above reaction, when `""_92^235U` is bombarded by neutron, it breaks up into two intermediate fragments which emit β-particles to achieve stable end products.
4) The energy released in fission first appears as K.E which gets converted into heat
in surrounding
5) Fission energy is being used in nuclear power projects for generation of electricity.
6) The uncontrolled fission process is used in atom bomb
APPEARS IN
RELATED QUESTIONS
Calculate the energy in fusion reaction:
`""_1^2H+_1^2H->_2^3He+n`, where BE of `""_1^2H`23He=7.73MeV" data-mce-style="position: relative;">=2.2323He=7.73MeV MeV and of `""_2^3He=7.73 MeV`
In a photon-electron collision ______.
(A) only total energy is conserved.
(B) only total momentum is conserved.
(C) both total energy and total momentum are conserved.
(D) both total momentum and total energy are not conserved
How long can an electric lamp of 100W be kept glowing by fusion of 2.0 kg of deuterium? Take the fusion reaction as
\[\ce{^2_1H + ^2_1H -> ^3_1He + n + 3.27 MeV}\]
Calculate the height of the potential barrier for a head on collision of two deuterons.
(Hint: The height of the potential barrier is given by the Coulomb repulsion between the two deuterons when they just touch each other. Assume that they can be taken as hard spheres of radius 2.0 fm.)
Write notes on Nuclear fusion
Explain the processes of nuclear fission and nuclear fusion by using the plot of binding energy per nucleon (BE/A) versus the mass number A
In a nuclear reaction
`"_2^3He + _2^3He -> _2^4He +_1^1H +_1^1H + 12.86 Me V` though the number of nucleons is conserved on both sides of the reaction, yet the energy is released. How? Explain.
Free 238U nuclei kept in a train emit alpha particles. When the train is stationary and a uranium nucleus decays, a passenger measures that the separation between the alpha particle and the recoiling nucleus becomes x in time t after the decay. If a decay takes place when the train is moving at a uniform speed v, the distance between the alpha particle and the recoiling nucleus at a time t after the decay, as measured by the passenger will be
During a nuclear fission reaction,
Show that the minimum energy needed to separate a proton from a nucleus with Zprotons and N neutrons is `ΔE = (M_(Z-1,N) + M_B - M_(Z,N))c^2`
where MZ,N = mass of an atom with Z protons and N neutrons in the nucleus and MB = mass of a hydrogen atom. This energy is known as proton-separation energy.
Consider the fusion in helium plasma. Find the temperature at which the average thermal energy 1.5 kT equals the Coulomb potential energy at 2 fm.
Calculate the Q-values of the following fusion reactions :-
(a) `""_1^2H + ""_1^2H → ""_1^3H + ""_1^1H`
(b) `""_1^2H + ""_1^2H → ""_2^3H + n`
(c) `""_1^2H + ""_1^3H → _2^4H + n`.
Atomic masses are `m(""_1^2H) = 2.014102 "u", m(""_1^3H) = 3.016049 "u", m(""_2^3He) = 3.016029 "u", m(""_2^4He) = 4.002603 "u".`
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
Why nuclear fusion reaction is also called thermo-nuclear reaction?
Write one balanced reaction representing nuclear fusion.
In a nuclear reactor, what is the function of a moderator?
In our Nature, where is the nuclear fusion reaction taking place continuously?
Explain in detail the four fundamental forces in nature.
Briefly explain the elementary particles present in nature.
