Advertisements
Advertisements
Question
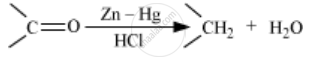
Write the reactions involved in the following reactions: Clemmensen reduction
Solution
Clemmensen reduction: The carbonyl group of aldehydes and ketones is reduced to CH2 group on treatment with zinc-amalgam and concentrated hydrochloric acid is know as Clemmensen reduction.
APPEARS IN
RELATED QUESTIONS
Complete the following reactions:
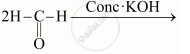
Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
Write the chemical equation for the reaction involved in Cannizzaro reaction.
An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
Complete the following reactions:
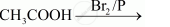
Complete the following reactions:
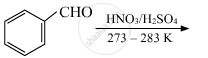
How will you convert acetone to acetone cyanohydrin?
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
\[\begin{array}{cc}
\ce{D}\phantom{........................}\\
|\phantom{.........................}\\
\ce{2D - C = O + OH^- ->[Cannizzaro] X and Y}
\end{array}\]
(Y is alcohol, D is deuterium)
X and Y will have the structure:
Convert the following:
Benzene to m-nitrobenzaldehyde
