Advertisements
Advertisements
Question
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Magnesium sulphite
Solution
\[\ce{MgSO3 + 2HCl -> MgCl2 + H2O + SO2}\]
APPEARS IN
RELATED QUESTIONS
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
State your observation When moist starch iodide paper is introduced into chlorine gas.
Name the gas that is produced in the given cases :
An action of dilute hydrochloride acid on sodium sulphide.
Explain why anhydrous \[\ce{HCl}\] is a poor conductor while aqueous \[\ce{HCl}\] is an excellent conductor.
Write an equation for the reaction of hydrochloric acid on zinc carbonate.
Fill in the blank:
On addition of silver nitrate to hydrochloric acid ___________ precipitate is formed which is soluble in ____________
Write balanced equation for the following reaction:
Copper oxide and dilute hydrochloric acid.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Write an equation for the reaction of hydrochloric acid on manganese (IV) oxide.
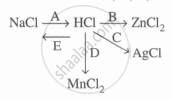
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.