Advertisements
Advertisements
Question
Write observation:
Lead nitrate solution is mixed with dilute hydrochloric acid and heated.
Solution
When dilute hydrochloric acid is added to the lead nitrate solution, white ppt of lead chloride appears.
\[\ce{Pb(NO3)2 + 2HCl -> PbCl2 ↓ + 2HNO3}\]
APPEARS IN
RELATED QUESTIONS
Dry hydrogen chloride gas can be collected by ________ displacement of air. (downward / upward)
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
Write an equation for the reaction of hydrochloric acid on zinc carbonate.
Explain why dilute hydrochloric acid cannot be concentrated by boiling beyond 22.2%.
What is the function of HCI in preparation of aqua-regia?
What is the property of concentrated sulphuric acid which allows it to be used in the preparation of hydrogen chloride and nitric acid?
Give a chemical test to distinguish between the following pair of compounds:
Sodium chloride solution and sodium nitrate solution.
Give a chemical test to distinguish between the following pair of compounds:
Hydrogen chloride gas and hydrogen sulphide gas
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Iron (II) sulphide
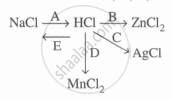
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.