Advertisements
Advertisements
Question
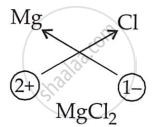
Write the chemical formula of the Magnesium chloride.
Solution
| Element Name | Magnesium |
| Element symbol | Mg |
| Charge | +2 |
| Valency | 1 Mg |
Hence formula: MgCl2
APPEARS IN
RELATED QUESTIONS
The element having atomicity 'four' is most likely to be :
Name the element in the compound and give the formula of the following compound:
Acetic acid
Write the formula of the following compound:
Potassium permanganate
Write the formula of the following compound:
Iron [II] chloride
Write the formula of the following compound:
Iron [III] hydroxide
Define the term Relative molecular mass. State why indirect methods are utilised to determine the absolute mass of an atom. Explain in brief the indirect method used.
State of the following formula of a compound is incorrect. Incorrectly write the correct formula.
NaZnO2
State of the following formula of a compound is incorrect. Incorrectly write the correct formula.
Mg(SO4)2
Complete the following crossword puzzle (Fig. 3.1) by using the name of the chemical elements. Use the data given in Table.
| Across | Down | ||
| 2 | The element used by Rutherford during his α–scattering experiment | 1 | A white lustrous metal used for making ornaments and which tends to get tarnished black in the presence of moist air |
| 3 | An element which forms rust on exposure to moist air | 4 | Both brass and bronze are alloys of the element |
| 5 | A very reactive non–metal stored under water | 6 | The metal which exists in the liquid state at room temperature |
| 6 | Zinc metal when treated with dilute hydrochloric acid produces a gas of this element which when tested with burning splinter produces a pop sound. | 8 | An element with symbol Pb |

