Advertisements
Advertisements
Question
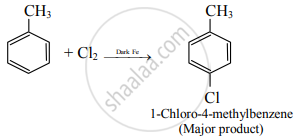
Write the structure and IUPAC name of the major product in the following reaction.
Solution
APPEARS IN
RELATED QUESTIONS
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2Cl + NaI ->[Acetone]}\\
|\phantom{..................}\\
\ce{CH3\phantom{...............}}
\end{array}\]
Write the structure and IUPAC name of the major product in the following reaction.
\[\ce{CH3 - CH2Br + SbF3 ->}\]
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH = CH2 + HBr ->[peroxide]}\\
|\phantom{........................}\\
\ce{CH3\phantom{.....................}}
\end{array}\]
Write the structure and IUPAC name of the major product in the following reaction.
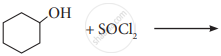
Give reason:
Alkyl halides are generally not prepared by free radical halogenation of alkanes.
Convert the following:
But-1-ene to n-butyl iodide
Name the reagent used to convert alkyl halide to ester.
Give reason. Though alkyl halides are moderately polar, they are insoluble in water.
What is Grignard reagent? How is it prepared? Why are they prepared under anhydrous condition?
Which of the following pairs of aryl halides cannot be prepared directly by electrophilic substitution?
Identify Sandmeyer's reaction from the following.
What is the name of reaction involving replacement of diazonium group by chloride using cuprous (I) salt?
Conversion of an alcohol into alkyl chloride by reacting with thionyl chloride is done in a medium of ____________.
What is the relative rate of SN2 reaction for (CH3)3C - Br?
An alkyl iodide on refluxing with aqueous KOH solution gave isopropyl alcohol. The structure of alkyl iodide could be:
Identify the name of reaction in which alkyl fluorides are prepared by heating alkyl bromide with metallic fluorides.
The conversion of ethyl bromide using sodium iodide and dry acetone, this reaction is know as ______.
Choose the false statement from the following.
How is alkyl halide converted into alcohol by using?
Aqueous NaOH (or KOH)
What is the advantage of alkyl halide using the thionyl chloride (SOCl2 ) method?
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
