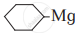Advertisements
Online Mock Tests
Chapters
2: Solutions
3: Ionic Equilibria
4: Chemical Thermodynamics
5: Electrochemistry
6: Chemical Kinetics
7: Elements of Groups 16, 17 and 18
8: Transition and Inner transition Elements
9: Coordination Compounds
▶ 10: Halogen Derivatives
11: Alcohols, Phenols and Ethers
12: Aldehydes, Ketones and Carboxylic acids
13: Amines
14: Biomolecules
15: Introduction to Polymer Chemistry
16: Green Chemistry and Nanochemistry
![Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 10 - Halogen Derivatives Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 10 - Halogen Derivatives - Shaalaa.com](/images/chemistry-english-12-standard-hsc_6:74400b801d4c44ef8e058ff9d9dfe964.jpg)
Advertisements
Solutions for Chapter 10: Halogen Derivatives
Below listed, you can find solutions for Chapter 10 of Maharashtra State Board Balbharati for Chemistry [English] 12 Standard HSC.
Balbharati solutions for Chemistry [English] 12 Standard HSC 10 Halogen Derivatives Exercises [Pages 231 - 233]
Choose the most correct option.
The correct order of increasing reactivity of C-X bond towards nucleophile in the following compounds is:
(I)
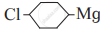
(II)
(III) (CH3)3 C-X
(IV) (CH3)2 CH-X
I < II < III < IV
II < I < III < IV
III < IV < II < I
IV < III < I < II
I < II < IV < III
\[\ce{CH3 - CH = CH2 ->[HI][Peroxide]}\]
The major product of the above reaction is, _______.
I - CH2 - CH = CH2
CH3 - CH2 - CH2I
\[\begin{array}{cc}
\ce{CH3-CH-CH3}\\
|\\
\ce{I}\end{array}\]\[\begin{array}{cc}
\ce{CH3-CH-CH2}\\
\phantom{.....}|\phantom{.....}|\\
\phantom{.......}\ce{I}\phantom{....}\ce{OH}
\end{array}\]
Choose the most correct option.
Which of the following is likely to undergo racemization during alkaline hydrolysis?
(I)
\[\begin{array}{cc}
\ce{CH3-CH-C2H5}\\
|\\
\ce{Cl}\end{array}\]
(II)

(III)
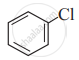
(IV)
\[\begin{array}{cc}
\ce{\phantom{.......}CH3}\\
\phantom{.....}|\\
\ce{CH3-CH}\\
\phantom{.....}|\\
\ce{\phantom{..........}CH2Cl}
\end{array}\]
Only I
Only II
II and IV
Only IV
Choose the most correct option.
The best method for the preparation of alkyl fluorides is _______.
Finkelstein reaction
Swartz reaction
Free radical fluorination
Sandmeyer's reaction
Identify the chiral molecule from the following.
1-Bromobutane
1,1-Dibromobutane
2,3-Dibromobutane
2-Bromobutane
An alkyl chloride on Wurtz reaction gives 2,2,5,5-tetramethylhexane. The same alkyl chloride on reduction with a zinc-copper couple in alcohol gives hydrocarbon with molecular formula \[\ce{C5H12}\]. What is the structure of alkyl chloride?
\[\begin{array}{cc}
\ce{CH3}\\
|\phantom{...}\\
\ce{CH3-C-CH2Cl}\\
|\phantom{...}\\
\ce{CH3}
\end{array}\]\[\begin{array}{cc}
\ce{CH3\phantom{..}}\\
|\phantom{.....}\\
\ce{CH3-C-CH2CH3}\\
|\phantom{.....}\\
\ce{Cl\phantom{....}}
\end{array}\]\[\begin{array}{cc}
\ce{CH3-CH2-CH-Cl}\\
\phantom{......}|\\
\ce{\phantom{.........}CH3}
\end{array}\]\[\begin{array}{cc}
\ce{CH3-CH-CH-CHCl}\\
|\phantom{.....}|\phantom{...}\\
\ce{\phantom{..}CH3} \ce{\phantom{..}CH3\phantom{..}}
\end{array}\]
Butanenitrile may be prepared by heating ______.
propanol with KCN
butanol with KCN
n-butyl chloride with KCN
n-propyl chloride with KCN
Choose the most correct option.
Choose the compound from the following that will react fastest by SN1 mechanism
1-Iodobutane
1-Iodopropane
2-Iodo-2 methylbutane
2-Iodo-3-methylbutane
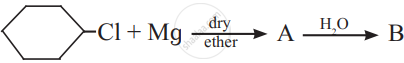
The product ‘B’ in the above reaction sequence is,
Choose the most correct option.
Which of the following is used as a source of dichlorocarbene?
tetrachloromethane
chloroform
iodoform
DDT
Write IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CH = C - CH - Br}\\
\phantom{......}|\phantom{....}|\\
\ce{\phantom{......}H3C} \ce{\phantom{...}CH3}
\end{array}\]
Write IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH2 - CH3}\\
|\phantom{......}|\phantom{.........}\\
\ce{Cl} \ce{\phantom{....}CH3\phantom{.......}}
\end{array}\]
Write IUPAC name of the following compound.
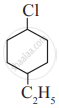
Write IUPAC name of the following compound.
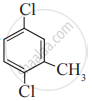
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2Cl + NaI ->[Acetone]}\\
|\phantom{..................}\\
\ce{CH3\phantom{...............}}
\end{array}\]
Write the structure and IUPAC name of the major product in the following reaction.
\[\ce{CH3 - CH2Br + SbF3 ->}\]
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH = CH2 + HBr ->[peroxide]}\\
|\phantom{........................}\\
\ce{CH3\phantom{.....................}}
\end{array}\]
Write the structure and IUPAC name of the major product in the following reaction.
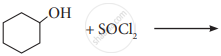
Write the structure and IUPAC name of the major product in the following reaction.
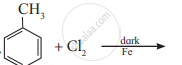
Identify chiral molecule/s from the following.
\[\begin{array}{cc}\ce{CH3 - CH - CH2 - CH3}\\|\phantom{.........}\\\ce{OH\phantom{.......}}\end{array}\]
\[\begin{array}{cc}\ce{CH3 - CH2 - CH - CH2 - CH3}\\
|\\\phantom{.}\ce{Br}\end{array}\]\[\ce{CH3 - CH2 - CH2 - CH2Br}\]
\[\begin{array}{cc}\ce{CH3 - CH - CH2 - CH3}\\
|\phantom{.........}\\\ce{CH3\phantom{......}}\end{array}\]
from the following pair would undergo SN2 faster from the other?
a. ![]() b.
b. ![]()
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Complete the following reaction giving major products.
\[\ce{CH3 - CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
Complete the following reaction giving major products.
\[\begin{array}{cc}\ce{CH3 - CH - CH3 ->[Red P/Br2] A ->[Ag2O/H2O]B}\\|\phantom{.........................}\\
\ce{OH\phantom{.......................}}\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Complete the following reaction giving major products.

Name the reagent used to bring about the following conversion.
Bromoethane to ethoxyethane
Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Name the reagent used to bring about the following conversion.
Ethyl bromide to ethyl isocyanide
Name the reagent used to bring about the following conversion.
Chlorobenzene to biphenyl
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Match the pairs.
| Column I | Column II |
| \[\begin{array}{cc}\ce{CH3CH - CH3}\\|\phantom{....}\\ \ce{X\phantom{....}}\end{array}\] |
vinyl halide |
| CH2 = CH - CH2X | alkyl halide |
| CH2 = CH - X | allyl halide |
| benzyl halide | |
| aryl halide |
Give reasons:
Haloarenes are less reactive than haloalkanes.
Give reason:
Alkyl halides though polar are immiscible with water.
Give reason:
Reactions involving Grignard reagent must be carried out under anhydrous condition.
Give reason:
Alkyl halides are generally not prepared by free radical halogenation of alkanes.
Distinguish between SN1 and SN2 mechanism of substitution reaction.
Explain optical isomerism in 2-chlorobutane.
Convert the following:
Propene to propan-1-ol
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
Convert the following:
But-1-ene to n-butyl iodide
Convert the following:
2-Chloropropane to propan-1-ol
Convert the following:
tert-Butyl bromide to isobutyl bromide
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
How will you bring about the following conversion?
Propene to 1-nitropropane
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Name the type of halogen derivative.
Observe the following and answer the question given below.

Comment on the bond length of C–X bond in it.
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Solutions for 10: Halogen Derivatives
![Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 10 - Halogen Derivatives Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 10 - Halogen Derivatives - Shaalaa.com](/images/chemistry-english-12-standard-hsc_6:74400b801d4c44ef8e058ff9d9dfe964.jpg)
Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 10 - Halogen Derivatives
Shaalaa.com has the Maharashtra State Board Mathematics Chemistry [English] 12 Standard HSC Maharashtra State Board solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Balbharati solutions for Mathematics Chemistry [English] 12 Standard HSC Maharashtra State Board 10 (Halogen Derivatives) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Balbharati textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry [English] 12 Standard HSC chapter 10 Halogen Derivatives are Classification of Halogen Derivatives, Nomenclature of Halogen Derivatives, Methods of Preparation of Alkyl Halides, Physical Properties, Optical Isomerism in Halogen Derivatives, Chemical Properties, Reaction with Active Metals, Uses and Environmental Effects of Some Polyhalogen Compounds, Nomenclature, Reactions of Haloalkanes - Elimination Reactions.
Using Balbharati Chemistry [English] 12 Standard HSC solutions Halogen Derivatives exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Balbharati Solutions are essential questions that can be asked in the final exam. Maximum Maharashtra State Board Chemistry [English] 12 Standard HSC students prefer Balbharati Textbook Solutions to score more in exams.
Get the free view of Chapter 10, Halogen Derivatives Chemistry [English] 12 Standard HSC additional questions for Mathematics Chemistry [English] 12 Standard HSC Maharashtra State Board, and you can use Shaalaa.com to keep it handy for your exam preparation.