Advertisements
Advertisements
Question
Observe the following and answer the question given below.

Comment on the bond length of C–X bond in it.
Solution
The bond length of C – X bond is expected to be shorter than C – X bond in haloalkanes.
APPEARS IN
RELATED QUESTIONS
from the following pair would undergo SN2 faster from the other?
a. ![]() b.
b. ![]()
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Convert the following:
tert-Butyl bromide to isobutyl bromide
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Name the type of halogen derivative.
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
The following mechanism has been proposed for the reaction of NO2 with F2 to form NO2F:
\[\ce{NO2_{(g)} + F2_{(g)} -> NO2F_{(g)} + F_{(g)} (slow)}\]
\[\ce{F_{(g)} + NO2_{(g)} -> NO2F_{(g)} (fast)}\]
The order of the reaction with respect to NO2(g)
Which of the following carbocation is the most stable?
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
Write the product formed when alkyl halide reacts with silver nitrite.
The major product formed when 2-bromobutane is treated with alcoholic KOH is ______.
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the questions given below:
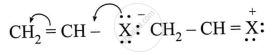
Name the type of halogen derivative.
Discuss β - elimination reaction.
Complete the following reaction sequences by writing the structural formulae of the organic compounds 'A', 'B' and 'C'.
\[\ce{2-Bromobutan ->[alc. KOH] A ->[][Br2] B ->[][NANH2] C}\]
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction, giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
