Advertisements
Advertisements
Question
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Solution
Compound (a) will undergo SN2 mechanism faster than (b).
APPEARS IN
RELATED QUESTIONS
from the following pair would undergo SN2 faster from the other?
a. ![]() b.
b. ![]()
Complete the following reaction giving major products.

Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Distinguish between SN1 and SN2 mechanism of substitution reaction.
Convert the following:
Propene to propan-1-ol
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
2-Chloropropane to propan-1-ol
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
How many metameric ethers are represented by the molecular formula C4H10O?
Which one is most reactive towards nucleophilic addition reaction?
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
Which of the following carbocation is the most stable?
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
The order of reactivities of the following alkyl halides for an SN2 reaction is ______.
The order of reactivity of the given haloalkanes towards nucleophiles is:
Observe the following and answer the question given below:
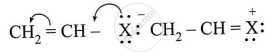
Comment on the bond length of C-X bond in it.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
