Advertisements
Advertisements
In Figure 1 below, a charge Q is fixed. Another charge q is moved along a circular arc MN of radius r around it, from the point M to the point N such that the length of the arc MN = l. The work done in this process is:
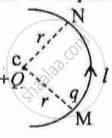
figure 1
Concept: undefined > undefined
If l3 and l2 represent angular momenta of an orbiting electron in III and II Bohr orbits respectively, then l3: l2 is :
Concept: undefined > undefined
Advertisements
With reference to the photoelectric effect, what is meant by threshold wavelength?
Concept: undefined > undefined
State any one property which is common to all electromagnetic waves.
Concept: undefined > undefined
Two metals A and B have work functions 4 eV and 6 eV respectively. Which metal has a lower threshold wavelength for photoelectric effect?
Concept: undefined > undefined
Calculate angular momentum of an electron in the third Bohr orbit of a hydrogen atom.
Concept: undefined > undefined
Plot a labelled graph of IVsl where Vs is stopping potential versus frequency f of the incident radiation.
Concept: undefined > undefined
State how will you use this graph to detennine the value of Planck's constant.
Concept: undefined > undefined
Draw energy level diagram for a hydrogen atom, showing the first four energy levels corresponding to n=1, 2, 3 and 4. Show transitions responsible for:
(i) Absorption spectrum of Lyman series.
(ii) The emission spectrum of the Balmer series.
Concept: undefined > undefined
If the frequency of the incident radiation is increased from 4 × 1015 Hz to 8 × 1015 Hz, by how much will the stopping potential for a given photosensitive surface go up?
Concept: undefined > undefined
The energy levels of an atom of a certain element are shown in the given figure. Which one of the transitions A, B, C, D or E will result in the emission of photons of electromagnetic radiation of wavelength 618.75 nm? Support your answer with mathematical calculations.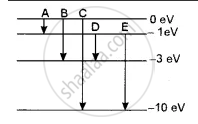
Concept: undefined > undefined
How are various lines of Lyman series formed? Explain on the basis of Bohr’s theory.
Concept: undefined > undefined
With reference to Semiconductor Physics,
Draw a labelled energy band diagram for a semiconductor.
Concept: undefined > undefined
If the radius of first electron orbit in hydrogen atom be r then the radius of the fourth orbit ill be ______.
Concept: undefined > undefined
The energy of an electron in an excited hydrogen atom is - 3.4 eV. Calculate the angular momentum of the electron according to Bohr's theory. (h = 6.626 × 10-34 Js)
Concept: undefined > undefined
Using Bohr's postulates derive the expression for the radius of nth orbit of the electron.
Concept: undefined > undefined
Ratio of longest to shortest wavelength in Balmer series is ______.
Concept: undefined > undefined
Light of wavelength 4000 Å is incident on two metals A and B. Which metal will emit photoelectrons, if their work functions are 3.8 e V and 1.6 e V respectively?
Concept: undefined > undefined
What is forbidden band?
Concept: undefined > undefined
An electric current (I) flowing through a metallic wire is gradually increased. The graph of heating power (P) developed in it versus the current (I) is ______.
Concept: undefined > undefined
