Advertisements
Advertisements
Match the following:
| 1 | Disaccharide | a | Lucas reagent |
| 2 | Carbylamine | b | Condensation polymer |
| 3 | Dacron | c | Obnoxious smell |
| 4 | Low spin complex `d^2sp^3` | d | Sucrose |
| 5 | Anhydrous `ZnCl_2 + conc.HCl` | e | Hexaamminecobalt(III)ion |
Concept: undefined > undefined
Name the important ore of silver. Write all the steps and reactions involved in the Cyanide process for the extraction of silver from its ore.
Concept: undefined > undefined
Advertisements
Answer the followiiig questions:
Identify the compounds A, B, C and D in the given reaction:
Concept: undefined > undefined
Identify the compounds A, B, C, D, E and F:
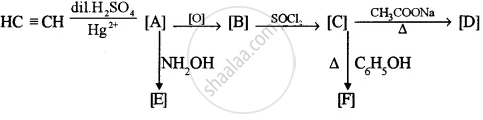
Concept: undefined > undefined
For the complexion of [Co(NH3)6]3+:
(i) State the hybridisation of the complex.
(ii) State the magnetic nature of the complex.
Concept: undefined > undefined
Give a reason for the following:
Fluorine gives only one oxide but chlorine gives a series of oxides.
Concept: undefined > undefined
Write the structures of all enantiomers possible for lactic acid.
Concept: undefined > undefined
Give balanced equations for the following reactions:
(i) Acetaldehyde is heated with hydroiodic acid in the presence of red phosphorous.
(ii) Calcium acetate is subjected to dry distillation.
(iii) Sodium ethoxide is treated with ethyl bromide.
(iv) Benzaldehyde is treated with sodium bisulphite.
Concept: undefined > undefined
Give balanced chemical equations for the following reactions:
(1) Acetaldehyde reacts with hydrogen cyanide.
(2) Acetone reacts with phenylhydrazine.
(3) Acetic acid is treated with ethanol and a drop of conc. H2SO4.
Concept: undefined > undefined
Give one chemical test each to distinguish between the following pairs of compounds:
(1) Acetone and benzaldehyde.
(2) Phenol and benzoic acid.
Concept: undefined > undefined
Choose the most correct option.
Which one of the following has the lowest acidity?
Concept: undefined > undefined
Identify the compounds [A], [B] and [C] in the following reaction.
\[\ce{C6H5NO2->[6{[H]}][Sn/HCl][A]->[HNO2 + HCl][273K-278K][B]->[C6H5NH2] [C]}\]
Concept: undefined > undefined
Identify the compounds [A], [B] and [C] in the following reaction.
\[\ce{CH3CH2NH2->[(1) HNO2][(2) P/Cl2][A]->[(1) KCN][(2) H2O/H+][B]->[(1) NH3/heat][(2) Br2/KOH][C]}\]
Concept: undefined > undefined
In non-ideal solution, what type of deviation shows the formation of maximum boiling azeotropes?
Concept: undefined > undefined
Define electrochemical cell
Concept: undefined > undefined
State Raoult’s law for the solution containing volatile components
Concept: undefined > undefined
Why does the cell potential of mercury cell remain constant throughout its life?
Concept: undefined > undefined
From the given cells:
Lead storage cell, Mercury cell, Fuel cell and Dry cell
Answer the following:
(i) Which cell is used in hearing aids?
(ii) Which cell was used in Apollo Space Programme?
(iii) Which cell is used in automobiles and inverters?
(iv) Which cell does not have long life?
Concept: undefined > undefined
What is meant by positive deviations from Raoult's law? Give an example. What is the sign of ∆mixH for positive deviation?
Concept: undefined > undefined
Define azeotropes.
Concept: undefined > undefined
