Advertisements
Advertisements
प्रश्न
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased ______.
विकल्प
both p and V of the gas will change.
only p will increase according to Charle’s law.
V will change but not p.
p will change but not V.
उत्तर
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased V will change but not p.
Explanation:
Consider the diagram where an ideal gas is contained in a cylinder, having a piston of mass M. Friction is absent.
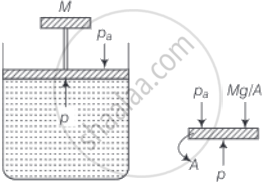
The pressure inside the gas will be P = Pa + Mg/A
Where, Pa = Atmosphere pressure
A = area of cross-section of the piston
Mg = Weight of piston
Hence, P = constant
When temperature increases as PV = nRT
⇒ Volume (V) increases at constant pressure.
APPEARS IN
संबंधित प्रश्न
It is said that the assumptions of kinetic theory are good for gases having low densities. Suppose a container is so evacuated that only one molecule is left in it. Which of the assumptions of kinetic theory will not be valid for such a situation? Can we assign a temperature to this gas?
When you come out of a river after a dip, you feel cold. Explain.
The average translational kinetic energy of gas molecules depends on ____________.
The average K.E. of hydrogen molecules at 27° C is E. The average K.E. at 627° C is ____________.
The average translational kinetic energy of a molecule in a gas becomes equal to 0.49 eV at a temperature about (Boltzmann constant = 1.38 x 10-23 JK-1) ____________.
Average kinetic energy of H2 molecule at 300K is 'E'. At the same temperature, average kinetic energy of O2 molecule will be ______.
A gas mixture consists of molecules of types A, B and C with masses mA > mB > mC. Rank the three types of molecules in decreasing order of rms speeds.
23Ne decays to 23Na by negative beta emission. Mass of 23Ne is 22.994465 amu mass of 23Na is 22.989768 amu. The maximum kinetic energy of emitted electrons neglecting the kinetic energy of recoiling product nucleus is ______ MeV.
The Q-value of a nuclear reaction and kinetic energy of the projectile particle, KP are related as ______.
According to the kinetic theory of gases, at a given temperature, molecules of all gases have the same ______.
