Advertisements
Advertisements
प्रश्न
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased ______.
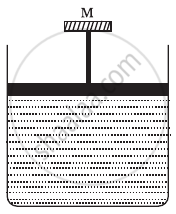
पर्याय
both p and V of the gas will change.
only p will increase according to Charle’s law.
V will change but not p.
p will change but not V.
उत्तर
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased V will change but not p.
Explanation:
Consider the diagram where an ideal gas is contained in a cylinder, having a piston of mass M. Friction is absent.
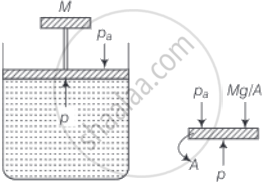
The pressure inside the gas will be P = Pa + Mg/A
Where, Pa = Atmosphere pressure
A = area of cross-section of the piston
Mg = Weight of piston
Hence, P = constant
When temperature increases as PV = nRT
⇒ Volume (V) increases at constant pressure.
APPEARS IN
संबंधित प्रश्न
The mean square speed of the molecules of a gas at absolute temperature T is proportional to
An adiabatic cylindrical tube of cross-sectional area 1 cm2 is closed at one end and fitted with a piston at the other end. The tube contains 0.03 g of an ideal gas. At 1 atm pressure and at the temperature of the surrounding, the length of the gas column is 40 cm. The piston is suddenly pulled out to double the length of the column. The pressure of the gas falls to 0.355 atm. Find the speed of sound in the gas at atmospheric temperature.
Answer in brief:
What will happen to the mean square speed of the molecules of a gas if the temperature of the gas increases?
At what temperature will oxygen molecules have same rms speed as helium molecules at S.T.P.? (Molecular masses of oxygen and helium are 32 and 4 respectively).
Answer in brief:
Compare the rms speed of hydrogen molecules at 127ºC with rms speed of oxygen molecules at 27ºC given that molecular masses of hydrogen and oxygen are 2 and 32 respectively.
If the density of nitrogen is 1.25 kg/m3 at a pressure of 105 Pa, find the root mean square velocity of nitrogen molecules.
What is the microscopic origin of temperature?
A molecule consists of two atoms each of mass 'm' and separated by a distance 'd'. At room temperature the average rotational kinetic energy is 'E', then its angular frequency is ______.
If a = 0. 72 and r = 0.24, then the value of t is ______.
2000 calories of radiant heat is incident on a body. If the body absorbs 550 calories of heat, find the coefficient of emmission of the body.
