Advertisements
Advertisements
प्रश्न
A gas mixture consists of molecules of types A, B and C with masses mA > mB > mC. Rank the three types of molecules in decreasing order of rms speeds.
उत्तर
As k = Boltzmann constant
T = Absolute temperature (same for all)
But mA > mB > mC
(vrms)A < (vrms)B < (vrms)C
∴ or (vrms)C < (vrms)B < (vrms)A
APPEARS IN
संबंधित प्रश्न
Can we define the temperature of (a) vacuum, (b) a single molecule?
The temperature and pressure at Simla are 15.0°C and 72.0 cm of mercury and at Kalka these are 35.0°C and 76.0 cm of mercury. Find the ratio of air density at Kalka to the air density at Simla.
Use R=8.314J K-1 mol-1
One mole of an ideal gas undergoes a process `P = (P_0)/(1+(V/V_0)^2` where `p_0` and `V_0` are constants . Find the temperature of the gas when `V=V_0` .
The weather report reads, "Temperature 20°C : Relative humidity 100%". What is the dew point?
Calculate the average molecular kinetic energy
- per kmol
- per kg
- per molecule
of oxygen at 127°C, given that the molecular weight of oxygen is 32, R is 8.31 J mol−1K−1 and Avogadro’s number NA is 6.02 × 1023 molecules mol−1.
The power radiated by a perfect blackbody depends only on its ______
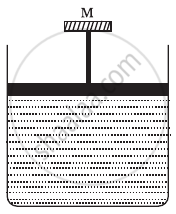
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased ______.
Explain why there is no atmosphere on moon.
The Q-value of a nuclear reaction and kinetic energy of the projectile particle, KP are related as ______.
Show that the average energy per molecule is proportional to the absolute temperature T of the gas.
